- 1Department of Gastroenterology, Qilu Hospital, Cheloo College of Medicine, Shandong University, Jinan, China
- 2Laboratory of Translational Gastroenterology, Qilu Hospital, Cheloo College of Medicine, Shandong University, Jinan, China
Background: Microbial dysbiosis is closely associated with visceral hypersensitivity and is involved in the pathogenesis of irritable bowel syndrome (IBS), but the specific strains that play a key role have yet to be identified. Previous bioinformatic studies have demonstrated that Fusobacterium is a shared microbial feature between IBS patients and maternal separation (MS)-stressed rats. In this study, we assessed the potential role of Fusobacterium nucleatum (F. nucleatum) in the pathogenesis of IBS.
Methods: Fecal samples of patients with diarrhea predominant-IBS (IBS-D) and healthy controls were obtained. An MS rat model was established to receive gavage of either F. nucleatum or normal saline. Visceral sensitivity was evaluated through colorectal distension test, and fecal microbiota was analyzed by 16S rRNA gene sequencing. F. nucleatum-specific IgA levels in fecal supernatants were assessed by western blotting. The antigen reacted with the specific IgA of F. nucleatum was identified by mass spectrometry and the construction of a recombinant Escherichia coli BL21 (DE3).
Results: IBS-D patients showed a lower Shannon index and a higher abundance of Fusobacterium. The F. nucleatum-gavage was shown to exacerbate visceral hypersensitivity in MS rats, with both the F. nucleatum-gavage and MS causing a decreased Shannon index and a clear segregation of fecal microbiota. In addition, specific IgA against F. nucleatum was detected in fecal supernatants of both the F. nucleatum-gavaged rats and the IBS-D patients. The FomA protein, which is a major outer membrane protein of F. nucleatum, was confirmed to react with the specific IgA of F. nucleatum in fecal supernatants.
Conclusion: Fusobacterium increased significantly in IBS-D patients, and F. nucleatum was involved in the pathogenesis of IBS by causing microbial dysbiosis and exacerbating visceral hypersensitivity in a colonization-independent manner. Meanwhile, F. nucleatum was found to induce an increase in specific secretory IgA through FomA.
Introduction
Irritable bowel syndrome (IBS) is a common functional gastrointestinal disorder that primarily manifests as abdominal pain and stool irregularities (Enck et al., 2016). Although IBS affects 11% of the global population (Lovell and Ford, 2012), its pathogenesis still remains unelucidated.
Due to the importance of the gut microbiota and viscera in the microbiome–gut–brain axis, both microbial dysbiosis and visceral hypersensitivity have been considered to be associated with the severity of IBS symptoms (Malinen et al., 2010; Simren et al., 2013, 2018). IBS patients often suffer from psychologically abnormal comorbidity (Bengtson et al., 2015) that can lead to the development of visceral pain sensation (Greenwood-Van Meerveld and Johnson, 2017). Moreover, fecal microbiota from IBS patients was observed to cause visceral hypersensitivity (Crouzet et al., 2013) and motility dysfunction when inoculated into germ-free mice (De Palma et al., 2017). However, a unique mechanism of microbially induced IBS has not been identified.
In a previous study, maternal separation (MS)-stressed rats were observed to form independent clusters that differed from the normal control group, and Fusobacterium was subsequently identified as one of the shared microbial features between IBS patients and MS-stressed rats in our previous research (Zhou et al., 2016). Thus, Fusobacterium may be closely related to the visceral hypersensitivity associated with IBS. Fusobacterium nucleatum (F. nucleatum), a typical strain of the genus Fusobacterium, is a gram-negative, obligate anaerobic bacterium that constitutively colonizes the oral mucosa (Aagaard et al., 2012; Segata et al., 2012). F. nucleatum has been suggested to contribute to the etiology of some gastrointestinal disorders, such as appendicitis, colon cancer, and inflammatory bowel disease (IBD) (Han, 2015). In addition, F. nucleatum has been shown to be responsive to stress hormones and is associated with pain and cold sensitivity in the oral cavity (Hahn et al., 1993; Massey et al., 1993; Sandrini et al., 2015). Nevertheless, an understanding of the effect of F. nucleatum in visceral hypersensitivity, its mechanism in the pathogenesis and/or manifestation of IBS symptoms, and the correlation between F. nucleatum and IBS symptoms has remained elusive. In this study, to confirm the role of F. nucleatum in the pathogenesis of IBS, the effects of F. nucleatum in the development of hypersensitivity and the presence of F. nucleatum-specific IgA in fecal samples was evaluated.
Materials and Methods
Participants
Patients with diarrhea predominant-IBS (IBS-D) (diagnosed as per the Rome IV criteria) and healthy controls (HC) aged 18–65 years were recruited from Qilu Hospital of Shandong University between August 2016 and March 2018. This study was approved by the ethics committees in Qilu Hospital of Shandong University. Patients suspected of being postinfectious IBS were excluded due to the heterogeneity in pathophysiological mechanisms. The exclusion criteria included pregnancy; lactation; previous surgeries within half a year; vaccination within 3 months; organic diseases or mental diseases (e.g., IBD, coeliac disease, anaphylactic diseases, and/or clinical psychiatric disorders); treatment with systemic corticosteroid, antipsychotics, and immunosuppressant agents; and taking antibiotics, probiotics, and laxatives within a month. The severity of symptoms was assessed by the IBS symptom severity scale (IBS-SSS) (Francis et al., 1997). The Self-rating Anxiety Scale (SAS) (Zung, 1971) and Self-rating Depression Scale (SDS) (Zung, 1965) were used to evaluate anxiety and depression. All subjects gave written informed consent.
Animal Maintenance and Modeling
Pregnant Sprague Dawley rats were purchased from Beijing HFK Bioscience Co., Ltd, and fed in the Experimental Animal Center of Shandong University. All animals were maintained under pathogen-free conditions (22 ± 2°C, 12-h light–dark cycle). All experiments performed on animals were approved by the Ethical and Institutional Animal Care and Use Committee of Qilu Hospital of Shandong University.
The experimental procedure is shown in Figure 1. The MS pups were isolated from dams for 3 h (from 9:00 to 12:00 am) between postnatal days 2 and 14 (O’Mahony et al., 2011). All pups were weaned on postnatal day 22. Male pups were preferred since female pups are subject to hormonal alterations. The pups received either F. nucleatum (ATCC 25586, 109 cfu, 1 ml/100 g) or normal saline (1 ml/100 g) once a week between the 4th and 8th weeks by oral gavage. Based on animal modeling, four groups were used in this study: group MS with F. nucleatum, the MS and F. nucleatum-gavage treatment group; group F. nucleatum, the normal-breeding and F. nucleatum-gavage treatment group; group MS, the MS and normal saline-gavage treatment group; and control group, the normal-breeding and normal saline-gavage treatment group. Fecal samples were collected at the end of weeks 3, 8, and 12. Visceral sensitivity was evaluated at week 12.
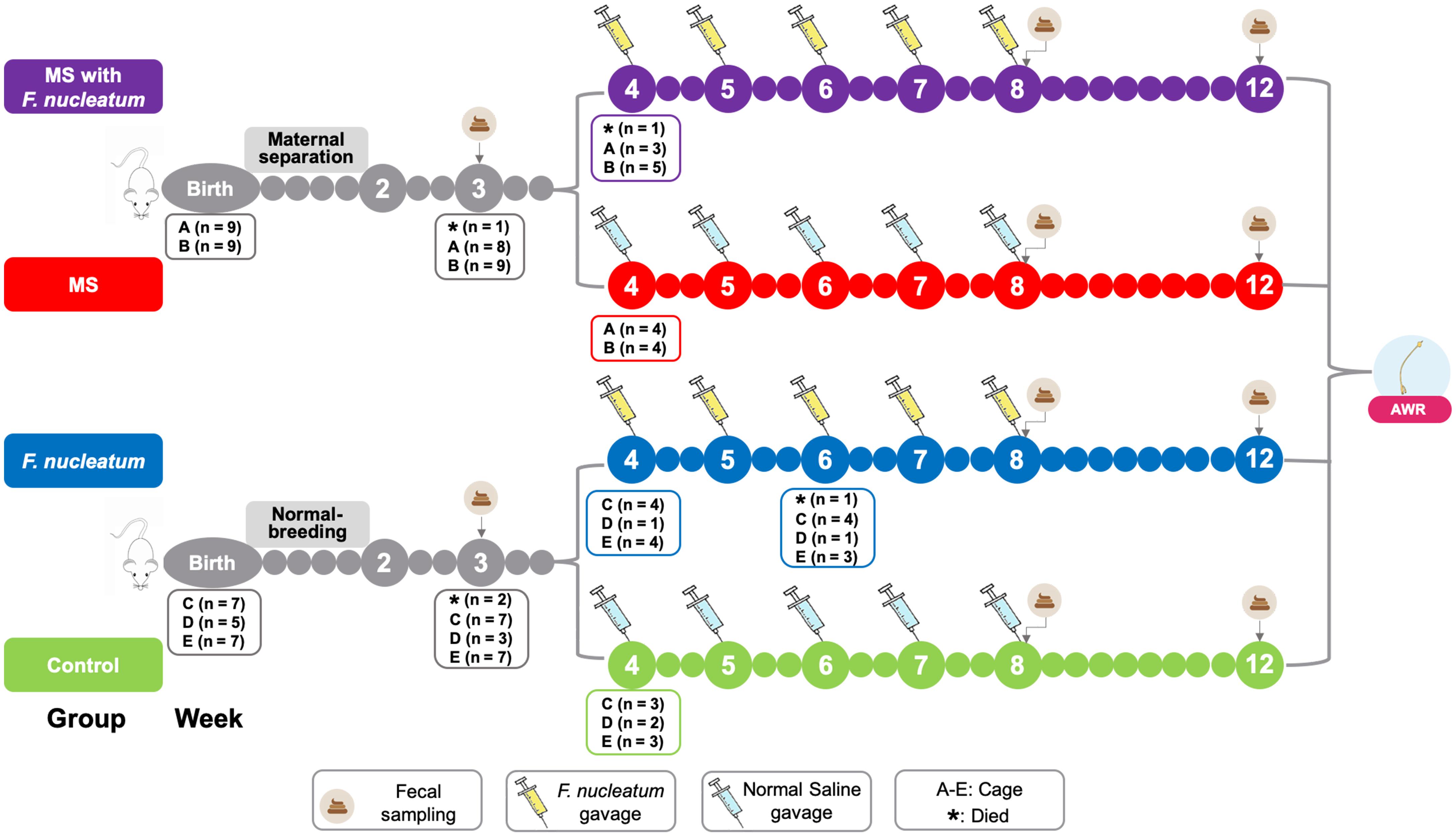
Figure 1. Study design. The treatment and the cohousing association of the experimental rats. MS, maternal separation; F. nucleatum, Fusobacterium nucleatum; AWR, abdominal withdraw reflex.
Bacterial Strains and Culture Conditions
F. nucleatum strain ATCC 25586 was purchased from the General Microbiological Culture Collection Center (Beijing, China). Escherichia coli (E. coli) BL21 (DE3) was purchased from TransGen Biotech (Beijing, China). F. nucleatum was cultured anaerobically at 37°C for 18 h in Brain Heart Infusion Broth (Qingdao Haibo Biotechnology Company, Qingdao, China, HB8297-4) before harvesting, while E. coli BL21 (DE3) strain was cultured aerobically at 37°C for 8 h in Luria-Bertani (LB) infusion broth.
Fecal Sample Collection, DNA Extraction, and 16S rRNA Gene Sequencing
Fecal samples collected from the IBS-D patients, HC, and rats were rapidly frozen in liquid nitrogen and stored at -80°C. These samples were then shipped to Majorbio (Shanghai, China) for high-throughput sequencing (Tang et al., 2018).
A FastDNA SPIN Kit (MP Biomedicals, Irvine, CA, United States) was used to extract stool DNA. Polymerase chain reaction (PCR) amplification of the V3–V4 regions of the bacterial 16S rRNA gene was performed with barcode-indexed primers (338F: ACTCCTACGGGAGGCAGCAG, 806R: ACTCCTACGGGAGGCAGCAG) and TransStart FastPfu DNA Polymerase (TransGen, Beijing, China) in an ABI GeneAmp instrument (ABI, United States). The amplicons were subsequently purified by gel extraction (AxyPrep DNA Gel Extraction Kit, Axygen, Union City, CA, United States) and quantified using a QuantiFluor-ST instrument (Promega, United States). The purified amplicons were pooled in equimolar concentrations, and paired-end sequencing was performed using an Illumina MiSeq System (Illumina, San Diego, CA, United States).
Visceral Hypersensitivity Evaluation
Visceral sensitivity was evaluated by colorectal distension (CRD) test (Xu et al., 2013). A urinary catheter (4 mm in diameter) was inserted into the colon via the anus. The balloon was placed 6 cm proximal to the anus and secured at the tail. The rats took approximately 10 min to adapt. Subsequently, the balloon was filled with saline to a constant volume. Each volume was repeatedly measured 5 times, 20 s at a time with a 5-min break. The abdominal withdraw reflex (AWR) scores were recorded as per the aforementioned methods (Al-Chaer et al., 2000). The overall visceral sensitivity was determined using the visceral hypersensitivity index (VHI), which involves obtaining the sum total of the AWR score from the balloon volume (Zhou et al., 2016).
Detection of F. nucleatum
F. nucleatum gene was detected by PCR. Total DNA was extracted from the stool of rats using TIANamp Stool DNA Kit (Tiangen, Beijing, China, DP328). The detection of F. nucleatum was performed using specific primers (Forward primer: 5′-CAACCATTACTTTAACTCTACCATGTTCA-3′; Reverse primer: 5′-GTTGACTTTACAGAAGGAGATTATGTAAAAATC-3′, Sangon Biotech Company, Shanghai, China) in a Mastercycler® pro S instrument (Eppendorf, Germany). Positive and negative controls were standard strains of F. nucleatum (ATCC 25586) and E. coli BL21 (DE3), respectively. The V4 region of the bacterial 16S rRNA gene was amplified by primers (515F: 5′-GTGCCAGCAGCCGCGGTAA-3′ and 806R: 5′-GGACTACCAGGGTATCTAAT-3′, Sangon Biotech Company, Shanghai, China) (Zhao et al., 2017) as internal control.
Specific IgA Detection and Antigen Identification
Fecal Supernatant (FSN) Collection
The fecal samples of the IBS-D patients, healthy volunteers, and rats were diluted in normal saline (1 g sample/7 ml normal saline) supplemented with 1% PMSF (Solarbio, Beijing, China, P0100) and homogenized on ice (Wang P. et al., 2016). The supernatants were collected after centrifugation (10,000 g, 4°C, 10 min), filtered using 0.22-μm micron syringe filters (Millex-GP, United States, SLGP033RB), and stored in a fridge at -80°C.
Detection of Total Protein Concentration and IgA Concentration
The total protein concentration of F. nucleatum, E. coli, and FSN was determined by BCA assay (BCA Protein Assay kit, ABP Biosciences, United States). For quantification of total IgA in FSN, the ELISA assay (IgA Rat Uncoated ELISA Kit with Plates, IgA Human Uncoated ELISA Kit with Plates, Invitrogen, United States) was performed in a 96-well flat-bottom plate. Both of the BCA and ELISA assays were performed according to the manufacturer’s instructions.
Specific IgA Detection via Western Blotting
The process was based on the protocols described previously (Wang H. F. et al., 2016; Donaldson et al., 2018). F. nucleatum or E. coli was collected, resuspended in normal saline, and the total protein was extracted by hypothermic ultrasonification. The F. nucleatum and E. coli proteins were denatured and separated by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE). The proteins were then transferred to polyvinylidene difluoride (PVDF) membranes. After blocking with 5% non-fat milk at room temperature for 5 h, the membrane strips were incubated at 4°C for 20 h with FSN from either human or rats that had been diluted to a specific concentration in normal saline. After washing, the PVDF membrane strips were incubated with either horseradish peroxidase (HRP)-labeled goat anti-human IgA alpha chain (1:5,000, Abcam, United Kingdom, ab97215) or HRP-labeled goat anti-rat IgA alpha chain (1:5,000, Abcam, United Kingdom, ab97185) for 60 min at room temperature. Immobilon Western Chemiluminescent HRP Substrate (Millipore, United States, WBKLS0100) was used to detect the bands.
Antigen Investigation Using Mass Spectrometry
The proteins were extracted from the gels after SDS-PAGE (10%), and the samples were treated (Qin and Zhang, 2002), cleaned, and digested using modified trypsin (Promega, United States) in the digestion buffer (100 mM ammonium bicarbonate, pH 8.5). Peptides were extracted using acetonitrile and dried down completely in a SpeedVac device (Thermo, United States). The dried samples were subsequently redissolved in 2% acetonitrile, 97.5% water, and 0.5% formic acid solution. The protein solution sample was first reduced by DL-dithiothreitol (DTT), and all cysteine residues were alkylated using iodoacetamide and cleaned either with desalting columns or by ethanol precipitation. The samples were then digested with sequencing grade modified trypsin in digestion buffer. The dissolved peptide samples were then analyzed with a NanoLC-ESI-MS/MS system, and the mass spectrometric data was used to search the NCBI NR protein database, after which the relative abundances of the proteins were calculated (Griffin et al., 2010).
Antigen Identification Using Recombination
The following procedures were performed to confirm that FomA (a major outer membrane protein of F. nucleatum) was recognized by the specific IgA (Liu et al., 2010). The sequence of the F. nucleatum fomA gene (GenBank Accession Number: X72582) was amplified using a forward PCR primer (5′-TTTC ATATGGAAGTTATGCCTGCACCTAC-3′) containing an NdeI site (Thermo Scientific, United States, FD0583) and a reverse PCR primer (5′-CGGCTCGAGTTAAGTAACTTTCATACC AG-3′) containing a XhoI site (Thermo Scientific, United States, FD0694). The primers were synthesized by the Sangon Biotech Company (Shanghai, China). The generated fragment was inserted into a pETDuet-1 vector that was subsequently transferred into E. coli BL21 (DE3) and plated onto LB agar medium containing ampicillin (50 μg/ml). Single colonies were selected and cultured in LB broth at 37°C for 8 h with shaking. The culture was diluted (1:100) with LB broth and incubated at 37°C, and isopropyl-β-D-thiogalactoside (IPTG) (Solarbio, Beijing, China, I8070) was added at a final concentration of 1 mM when the optical density at 620 nm reached 0.5. After induction, bacteria were harvested by centrifugation (6,000 × g, 4°C, 5 min) and then washed twice with normal saline. The total protein was extracted by hypothermic ultrasonification, after which SDS-PAGE was used to assess FomA expression, and then the reaction between the recombinant FomA and specific IgA of F. nucleatum in FNS was detected by western blotting as described above.
Statistics and Taxonomy Quantification
The Mann–Whitney non-parametric test, Kruskal–Wallis non-parametric test and Spearman correlation were used to assess differences among the groups and correlations between variables. SPSS (version 24.0, IBM, United States) was used to analyze data. A 95% confidence level, i.e., P < 0.05 was used.
The raw sequences were processed using Usearch version 8.1.1861_win32. The raw reads were dereplicated, and the singletons or chimeras were removed. The sequences were then clustered into operational taxonomic units (OTUs) based on a 97% similarity and analyzed using the RDP classifier Bayes algorithm to calculate the community composition at each taxonomic level. The OTU tables were imported into Mothur v 1.38.1 (Schloss et al., 2009) for the rarefaction curves and diversity (Shannon index) analyses. The specific taxa were identified by the linear discriminant analysis effect size (LEfSe) analysis with the value of the Kruskal–Wallis rank-sum test set to 0.05 and an LDA cutoff value of 2.5. The non-metric multi-dimensional scaling (NMDS) analysis and redundancy analysis (RDA) were performed in Mothur and plotted in R v 3.2.3 (R Core team, 2015).
Results
Fusobacterium Is a Microbial Biomarker of IBS-D
A total of 71 IBS-D patients and 39 HC were recruited. Demographics between IBS-D patients and HC were well balanced (Table 1).
A total of 110 fecal samples were analyzed using a MiSeq PE 300, with 5,530,955 valid reads obtained. In the analysis, 4,657,712 reads were used after noise reduction and removal of low-quality reads and chimeras. Subsequently, the reads were analyzed using Usearch and the RDP database, resulting in the identification of 2,425 OTUs.
The microbial diversity was compared by calculating the Shannon index. IBS-D patients showed a significantly lower Shannon index (P = 0.047, Mann–Whitney test) (Figure 2A). Furthermore, the microbial community structure was evaluated by NMDS analysis of beta diversity. IBS-D patients showed different microbial structures from healthy controls (P = 0.002, Adonis) (Figure 2B). Based on the observed OTU distributions, possible microbial biomarkers associated with IBS-D were determined by LEfSe analysis with the value of the Kruskal–Wallis rank-sum test set to 0.05 and an LDA cutoff value of 2.5 (Figure 2C, Supplementary Table 1 and Figure 1). The genus Faecalibacterium, genus Ruminococcus, genus Subdoligranulum, genus Dorea, etc., were highly enriched in HC, and in IBS-D patients, the genus Prevotella, genus Streptococcus, genus Roseburia, genus Fusobacterium, etc., were significantly enriched (Supplementary Table 1 and Figure 1). From these, all of the phylum Fusobacteria, class Fusobacteria, order Fusobacteriales, family Fusobacteriaceae, and genus Fusobacterium were shown to be possible microbial biomarkers of IBS-D patients (Figure 2C, Supplementary Table 1 and Figure 1). The abundance of Fusobacterium in the IBS-D patients was shown to remarkably higher than that observed in HC (P < 0.001, Mann–Whitney test) (Figure 2D). In addition, the abundance of Fusobacterium in IBS-D patients was positively correlated with SDS values (P = 0.034, rho = 0.252, Spearman correlation) (Supplementary Figure 2).
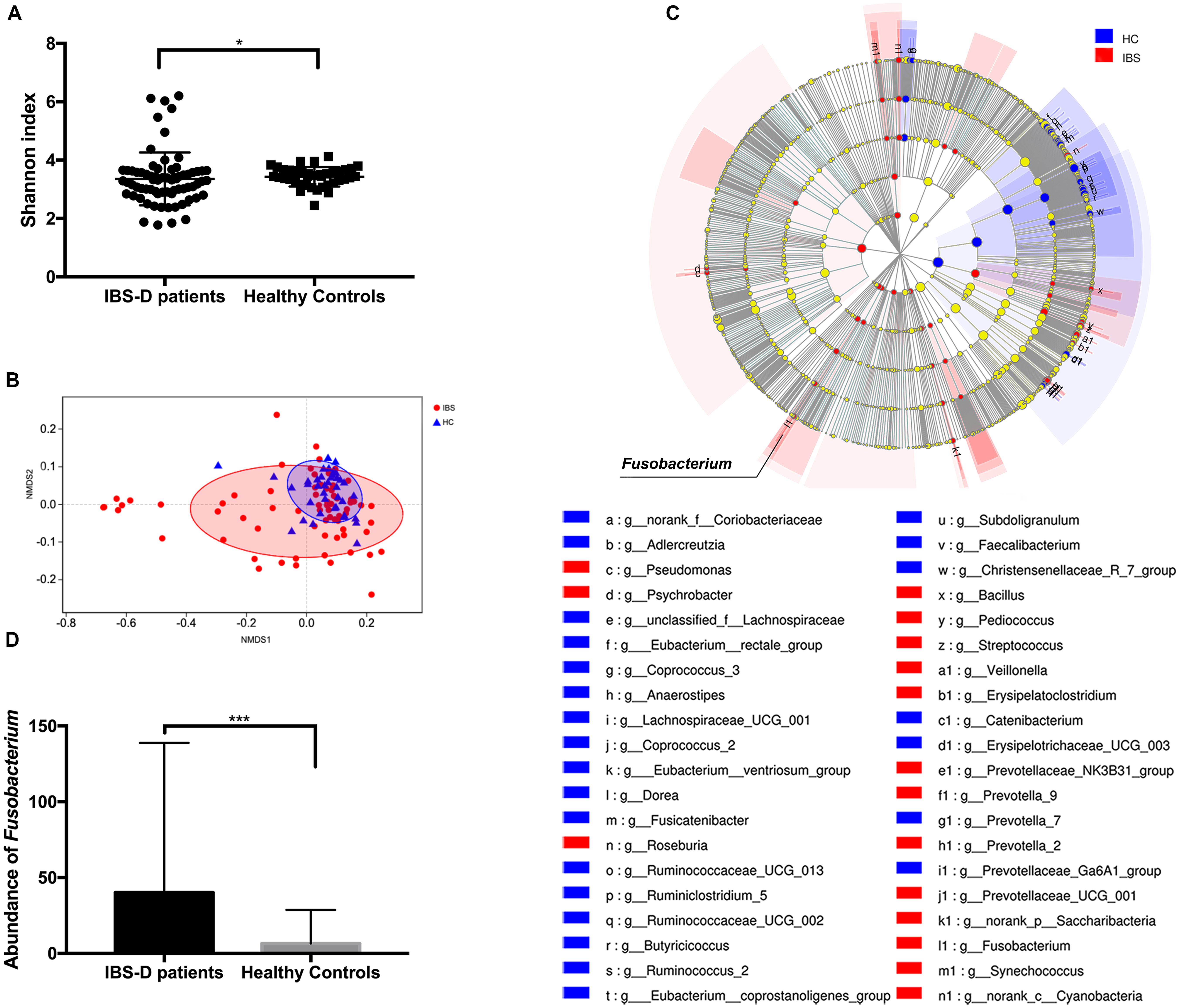
Figure 2. Intestinal microbiota characteristics in patients with diarrhea predominant-irritable bowel syndrome (IBS-D) and healthy controls (HC). (A) Shannon index between IBS-D patients and HC. (B) The non-metric multi-dimensional scaling (NMDS) analysis of IBS-D patients and HC. (C) The linear discriminant analysis effect size analysis with the value of the Kruskal–Wallis sum-rank test set to 0.05 and an LDA cutoff value of 2.5. (D) Abundance of F. nucleatum between IBS-D patients and HC. *P < 0.05, ***P < 0.001. IBS, IBS-D patients; HC, healthy controls.
F. nucleatum Causes the Intestinal Microbial Dysbiosis in Rats
A total of 96 fecal samples were analyzed using MiSeq PE 300, with 3,645,585 valid reads obtained. After noise reduction and removal of low-quality reads and chimeras, 3,008,922 reads were used. Subsequently, the reads were analyzed using the Usearch and the RDP database, with 1,239 OTUs obtained.
Microbial Diversity and NMDS Analysis
The microbial diversity was compared using the Shannon index, Sobs value, and Chao_1 value. Results showed that at week 3, there was no significant difference of Shannon index between the MS rats and normal-breeding rats (χ2 = 2.27, df = 1, P = 0.13, KW) (Figure 3A). The Shannon index of the control group was higher than that of the other three groups at week 8 (χ2 = 10.2926, df = 3, P = 0.02, KW) (Figure 3B), while group F. nucleatum had the highest Shannon index at week 12 (χ2 = 10.1278, df = 3, P = 0.02, KW) (Figure 3C). Moreover, MS rats had a lower Sobs value than normal-breeding rats at week 3 (χ2 = 5.114, df = 1, P = 0.024, KW) (Supplementary Figure 3A). The control group showed a significantly higher Sobs value than the other three groups at week 8 (χ2 = 11.747, df = 3, P = 0.008, KW) (Supplementary Figure 3B), while there was no clear difference among the four groups at week 12 (χ2 = 0.054, df = 3, P = 0.997, KW) (Supplementary Figure 3C). For the Chao_1 value, no difference was found at week 3 (χ2 = 2.876, df = 1, P = 0.090, KW) (Supplementary Figure 3D), and all the three treatment groups had a lower Chao_1 value than the control group at week 8 (χ2 = 12.847, df = 3, P = 0.005, KW) (Supplementary Figure 3E), whereas at week 12, the difference among groups was unapparent (χ2 = 0.114, df = 3, P = 0.990, KW) (Supplementary Figure 3F). These results demonstrated that both the MS and F. nucleatum-gavage could decrease the diversity of intestinal microbiota, with the F. nucleatum-gavage having a particularly lasting effect.
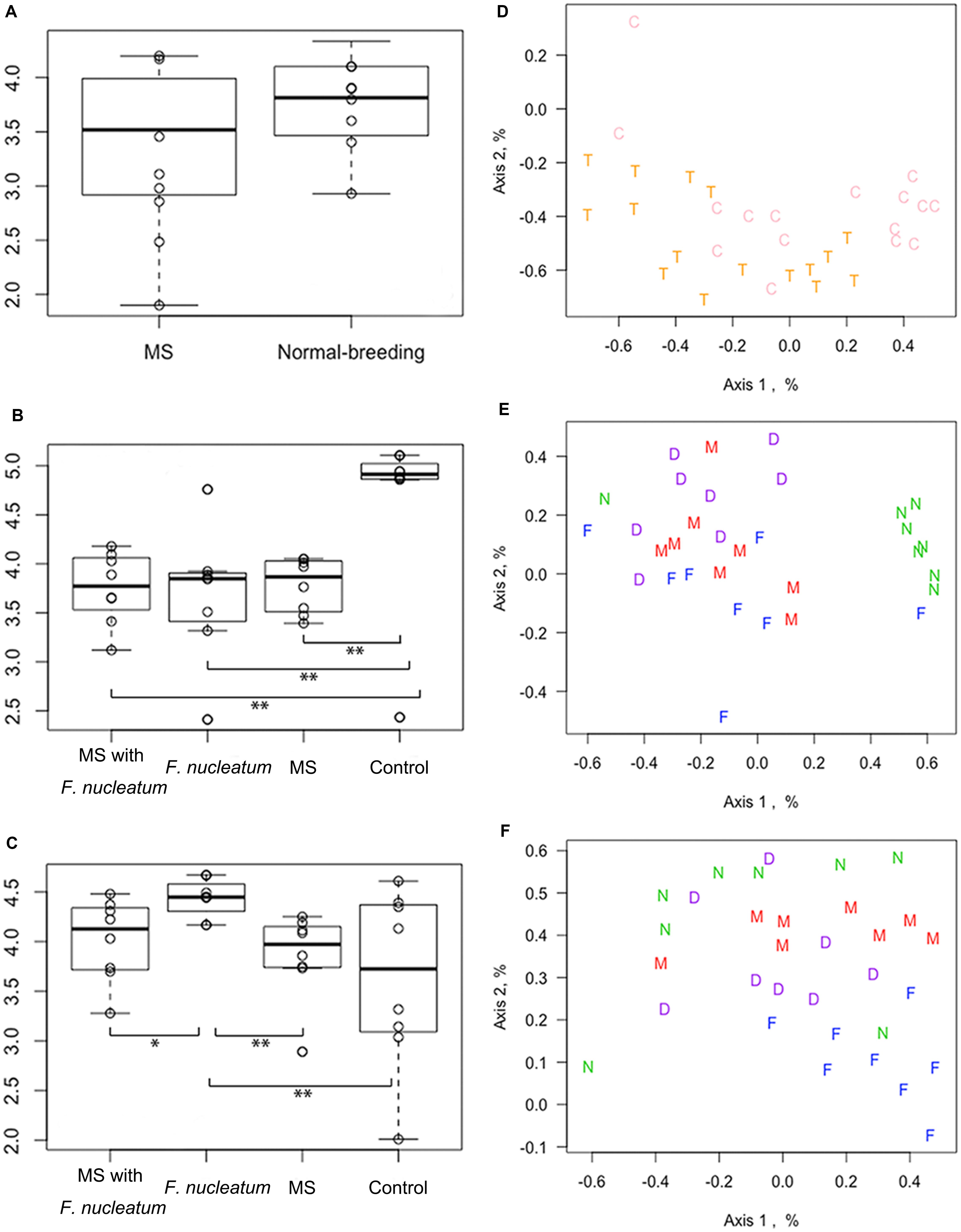
Figure 3. Microbial diversity and non-metric multi-dimensional scaling (NMDS) analysis. The diversity was analyzed by Shannon index at weeks 3 (A), 8 (B), and 12 (C). The NMDS was analyzed at week 3 (D), 8 (E), and 12 (F). *P < 0.05, **P < 0.01. MS, maternal separation; C, normal-breeding rats; T, MS rats; D, group MS with F. nucleatum; M, group MS; F, group F. nucleatum; N, control group.
The OTU data was clustered through an NMDS analysis. At week 3, no visible distinction was observed (Figure 3D). However, the control group had notably separated from the other three groups at week 8 (Figure 3E), although this separation was not obvious at week 12 (Figure 3F). Microbial dysbiosis was further evaluated by assessing the effect of treatments and cohousing using the Adonis and Anosim test (Supplementary Table 2). At week 3, the fecal samples of the MS rats were distinct from that of normal-breeding rats (P < 0.01). A significant difference also existed across the four groups at both weeks 8 and 12 (P < 0.001). No significant difference in cohousing was observed.
Reduced Colonization of Dominant OTUs
A reduced colonization of dominant OTUs was observed from the rank abundance curve. No notable differences were observed at week 3 (Figure 4A), whereas at week 8, the curve of the control group was distinct from those of the other groups (Figure 4B). Notably, the first two ranks of the group F. nucleatum were less than those of the other three groups at week 12 (Figure 4C). The relative amounts of the most abundant OTU at week 12 were then compared, showing that that of group F. nucleatum was lesser than those of the other groups (P = 0.01; P = 0.03; P = 0.008, Mann–Whitney test) (Figure 4D). This result indicated that the species in group F. nucleatum were more evenly distributed, consistent with the result that the diversity of group F. nucleatum was the highest at week 12 as described above.
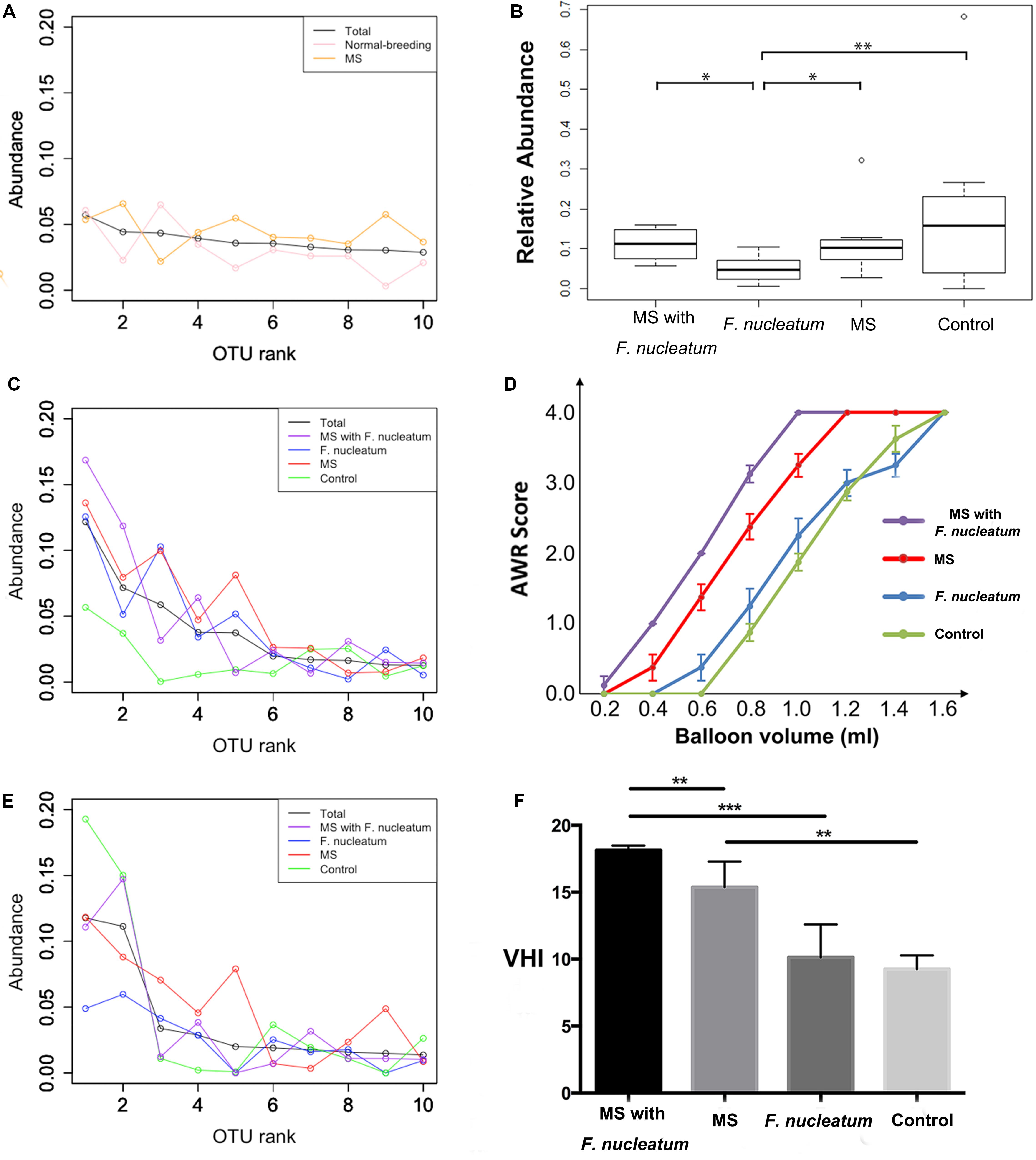
Figure 4. Rank-abundance curve and visceral sensitivity evaluation. For rank-abundance curves, the OTU ranks at the X-axis were ordered by the mean abundance of total samples, and the curve of each group was referred to the same ranks of total samples. Curves were plotted at weeks 3 (A), 8 (B), and 12 (C). (D) The relative amount of the most abundant OTU at week 12. (E) Abdominal withdraw reflex (AWR) score. (F) Visceral hypersensitive index (VHI). *P < 0.05, **P < 0.01, ***P < 0.001. MS, maternal separation.
F. nucleatum Exacerbates Visceral Hypersensitivity in MS Rats
The visceral sensitivity of each group was evaluated by determining AWR scores based on graded CRD (Figure 4E). The VHI (Zhou et al., 2016) from the sum of the AWR scores at 0.4, 0.6, 0.8, 1.0, 1.2, and 1.4 ml were calculated (Figure 4F). Significant differences in the VHI values were observed among four groups (χ2 = 20.313, df = 8, P = 0.009, KW). Group MS with F. nucleatum and group MS had higher VHI values than group F. nucleatum and the control group (P < 0.001 and P = 0.001, respectively, Mann–Whitney test). The VHI of group MS with F. nucleatum was higher than that of group MS (P = 0.003, Mann–Whitney test). These results showed that F. nucleatum-gavage exacerbated visceral hypersensitivity in MS rats.
F. nucleatum Does Not Colonize but Imprints the Immune System
Detection of Specific IgA Against F. nucleatum in Rat FSN and RDA
F. nucleatum was not detected from 16s rRNA gene sequencing or PCR (Supplementary Figure 4). Microbial antigens were previously shown to induce mucosal immunity where microbial neutralization was achieved by secretory IgA (Macpherson and Uhr, 2004; Konrad et al., 2006). No significant difference was tested in the total protein concentration of FSN among groups at week 3, week 8, or week 12 (Supplementary Figure 5), and for the IgA concentration in FSN, there was no significant difference among groups by time points (Supplementary Figure 6). Thus, F. nucleatum-specific IgA in the FSNs of rats was detected by western blotting. Ponceau S staining was used as loading control (Figure 5D). No obvious reactive band was observed at week 3 (Figure 5A), whereas two strong bands with molecular masses of 40 and 130 kDa were detected at week 8 in most samples of group MS with F. nucleatum and group F. nucleatum. In contrast, two weak bands at the same positions were detected in some of the samples from group MS and the control group (Figure 5B). These two bands were also observed in most samples from group MS with F. nucleatum and group F. nucleatum at week 12 but were almost invisible in group MS and the control group (Figure 5C). Additionally, the results of a parallel experiment showed that the bands observed using E. coli BL21 (DE3) occurred at different positions, confirming the specificity of IgA toward F. nucleatum (Figure 7H). The proportional signals of these two reactive bands reflected the levels of F. nucleatum-specific IgA in FSN. After quantifying and comparing the proportional signals of these two bands (Donaldson et al., 2018), no differences were observed in either the 130- or 40-kDa bands at week 3 (Figure 5E). At week 8, there were significant differences among groups at both 130 kDa (χ2 = 13.943, df = 3, P = 0.003, KW) and 40 kDa (χ2 = 22.259, df = 3, P < 0.001, KW) (Figure 5F), at week 12 these differences were still significantly different at both 130 kDa (χ2 = 21.838, df = 3, P < 0.001, KW) and 40 kDa (χ2 = 18.219, df = 3, P < 0.001, KW) (Figure 5G). Thus, F. nucleatum protein was shown to stimulate the production of specific IgA in F. nucleatum-gavaged rats, and this effect was relatively persistent.
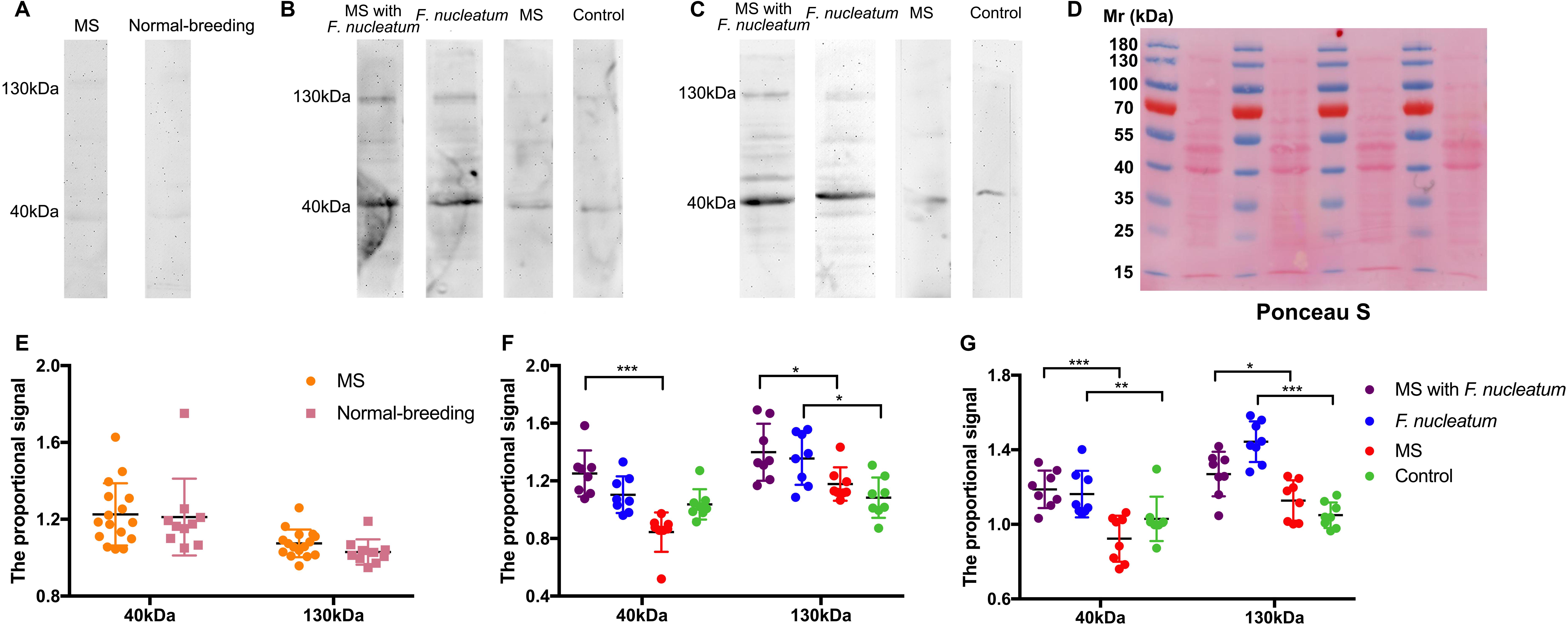
Figure 5. Detection of specific IgA against F. nucleatum in fecal supernatants (FSN) of rats. The western blotting was used to detect the IgA against F. nucleatum at weeks 3 (A), 8 (B), and 12 (C). (D), Ponceau S staining was used as loading control. The quantified proportional signals from reactive bands at 40 and 130 kDa were calculated at weeks 3 (E), 8 (F), and 12 (G). *P < 0.05, **P < 0.01, ***P < 0.001. MS, maternal separation.
The association between the levels of F. nucleatum-specific IgA in FSN and the microbial community was analyzed by RDA. As shown in Figure 6, the mean intensity showed the proportional signals, which at 130 and 40 kDa were crucial attributes that influenced the bacterial community. In particular, the mean intensity of the 130 and 40 kDa at weeks 8 and 12 was positively correlated with the microbial communities of group MS with F. nucleatum and group F. nucleatum, both of which received the F. nucleatum-gavage, whereas group MS and the control group exhibited a negative correlation (Figures 6B,C). The relationship between the levels of F. nucleatum-specific IgA in FSN and the abundance of specific bacterial taxa was analyzed. According to the Spearman correlation analysis, the mean intensity of the 130-kDa band at week 8 negatively correlated with the abundance of an unclassified OTU (P = 0.010, rho = -0.450, Figure 6D). The mean intensity of the 130-kDa band at week 12 correlated positively with the abundance of an OTU belonged to Clostridiales (P = 0.003, rho = 0.513, Figure 6E), but negatively correlated with the abundance of an OTU belonging to Enterococcaceae (P = 0.0003, rho = -0.599, Figure 6F). Additionally, the mean intensity of the 40-kDa band at week 12 negatively correlated with the abundance of an OTU belonging to Lactobacillus (P = 0.032, rho = -0.382, Figure 6G).
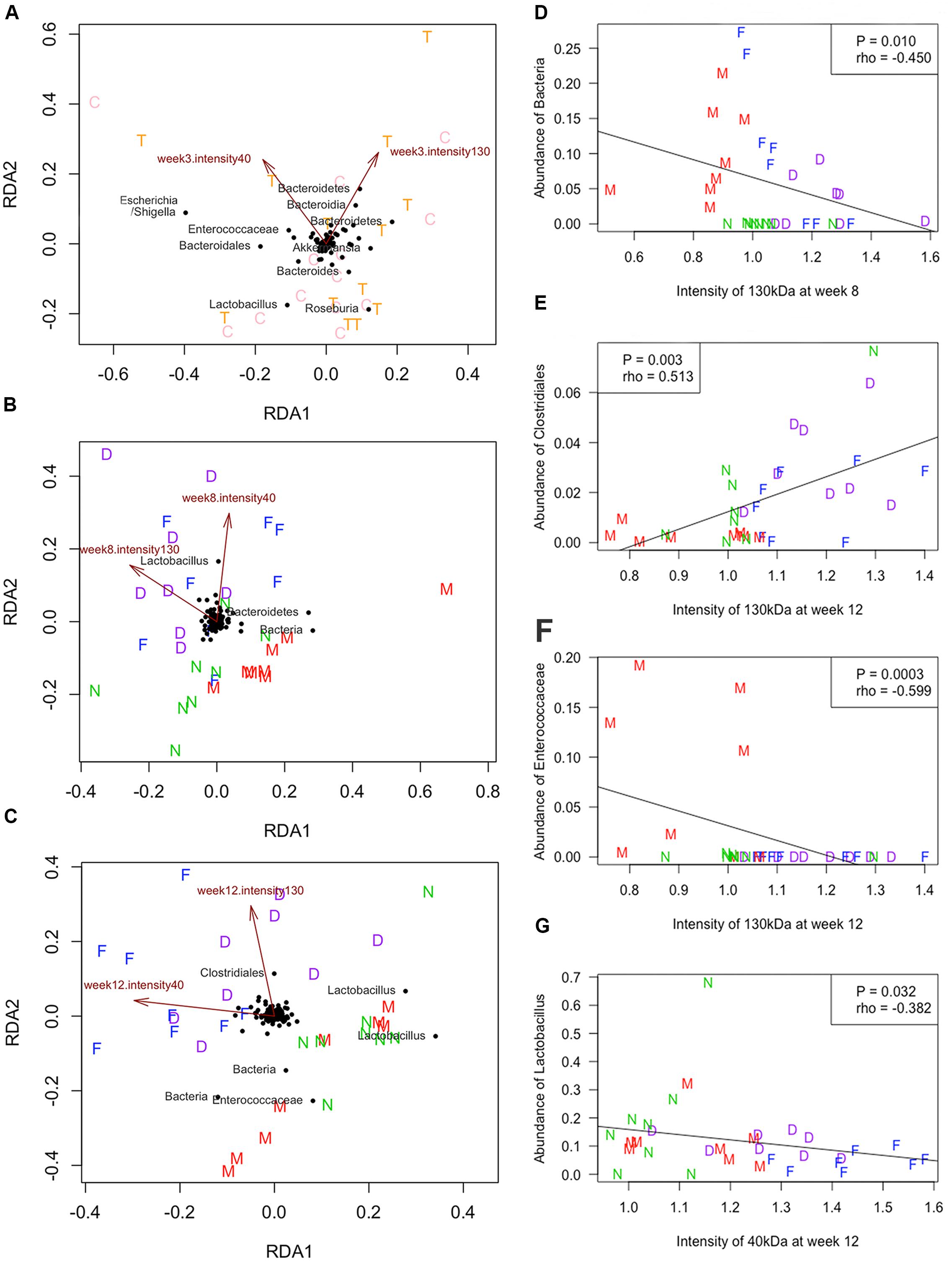
Figure 6. Redundancy analysis (RDA) and Spearman correlation. The RDA was performed at weeks 3 (A), 8 (B), and 12 (C). The quantified proportional signals from reactive bands at 130 and 40 kDa were used as environmental variables. The Spearman correlation was calculated between the quantified proportional signals at 130 kDa and abundance of an unclassified OTU at week 8 (D), the signals of 130 kDa and abundance of the OTU belonged to Clostridiales (E) or Enterococcaceae (F) at week 12, and signals of 40 kDa and abundance of the OTU belonged to Lactobacillus at week 12 (G). C, normal-breeding rats; T, maternal separation (MS) rats; D, group MS with F. nucleatum; M, group MS; F, group F. nucleatum; N, control group.
Detection of F. nucleatum-Specific IgA in Human FSN
To further confirm the association of F. nucleatum-specific IgA with IBS, the FSNs of seven IBS-D patients and five HC were investigated because of their sufficient fecal samples and complete clinical data. The patient characteristics are shown in Supplementary Table 3. No significant differences in age (P = 0.530, Mann–Whitney test) and body mass index (BMI) (P = 0.639, Mann–Whitney test) were observed. The IBS-D patients showed more notable anxiety and depression based on SAS (P = 0.003, Mann–Whitney test) and SDS (P = 0.003, Mann–Whitney test). The severity of intestinal symptoms was assessed using the IBS-SSS, with all IBS-D patients exhibiting severe IBS (total score >300).
There was no significant difference between IBS-D patients and HC in neither FSN total protein concentration nor FSN IgA concentration (Supplementary Figure 7). The detection of F. nucleatum-specific IgA was performed by western blotting. Ponceau S staining was used as loading control (Figure 7A). Two strong reactive bands at 130 and 40 kDa were observed using the FSN of IBS-D patients, whereas the use of the FSN from HC produced bands with relatively weak intensities (Figure 7A). This result was similar to those observed using rats. The quantified proportional signals from reactive bands were significantly higher in IBS-D patients at both 40 kDa (P = 0.003, Mann–Whitney test) (Figure 7B) and 130 kDa (P = 0.005, Mann–Whitney test) (Figure 7C). Furthermore, SAS was positively correlated with the quantified proportional signal for both the 130-kDa (P = 0.028, rho = 0.629, Spearman correlation) (Figure 7D) and 40-kDa bands (P = 0.003, rho = 0.783, Spearman correlation) (Figure 7E). Similarly, SDS exhibited a positive correlation with both the signals of the 130-kDa (P = 0.029, rho = 0.628, Spearman correlation) (Figure 7F) and 40-kDa bands (P = 0.018, rho = 0.667, Spearman correlation) (Figure 7G). Therefore, the specific IgA against F. nucleatum was suggested as a symptom-associated antibody in IBS-D patients.
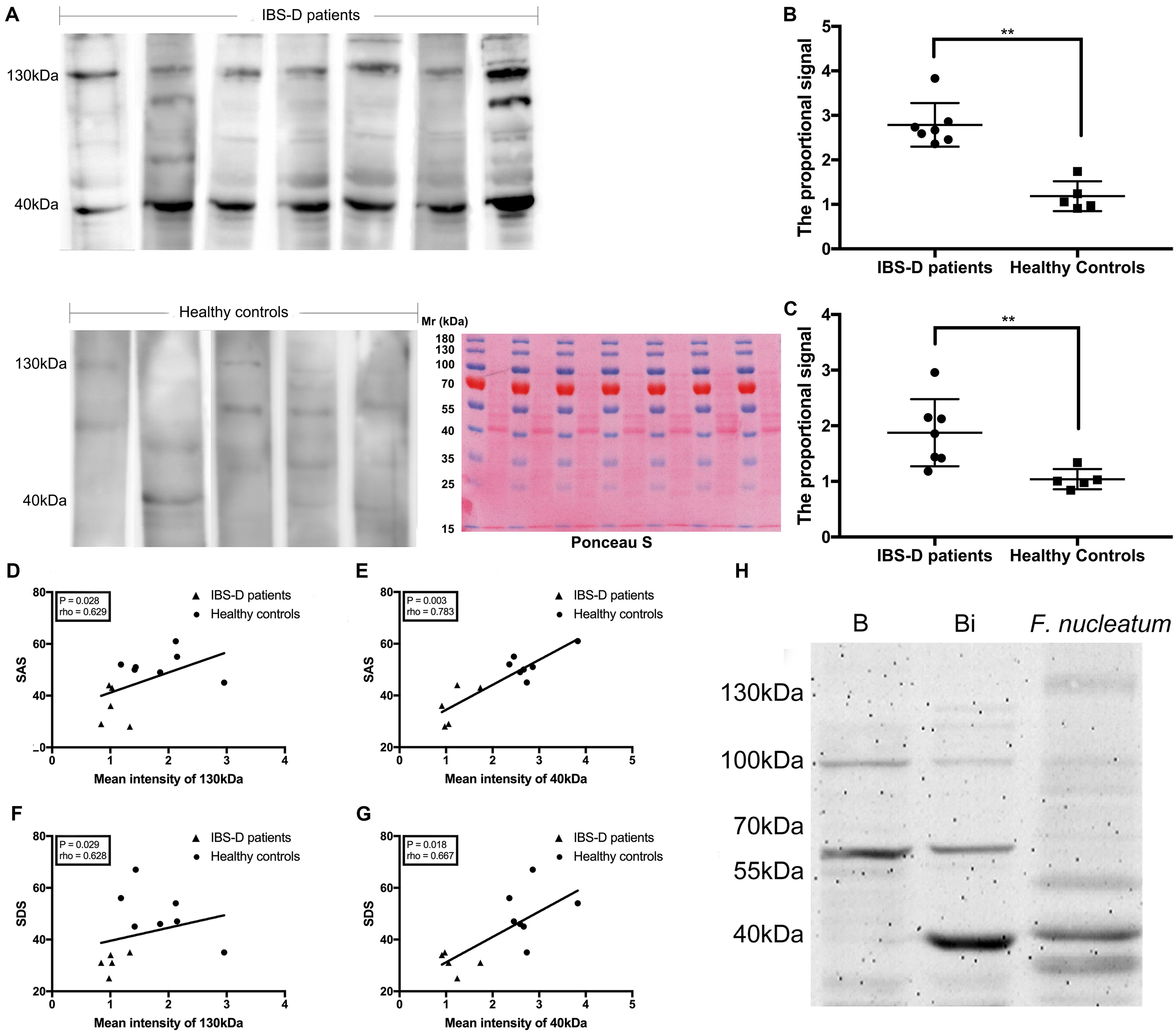
Figure 7. Detection of specific IgA against F. nucleatum in fecal supernatants (FSN) of patients and identification of FomA. The IgA against F. nucleatum was detected by western blotting, and Ponceau S staining was used as loading control (A). The quantified proportional signals from reactive bands were evaluated at 40 (B) and 130 kDa (C). The Spearman correlation was calculated between the Self-rating Anxiety Scale (SAS) and the quantified proportional signals from band at 130 (D) and 40 kDa (E), Self-rating Depression Scale (SDS), and signals from band at 130 (F) and 40 kDa (G). (H) The specific IgA in the FSN against E. coli BL21 (DE3) with or without expression of FomA gene. **P < 0.01. B: E. coli BL21 (DE3) without expression of FomA gene; Bi: E. coli BL21 (DE3) with expression of the FomA gene.
Identification of a F. nucleatum-Specific Antigen
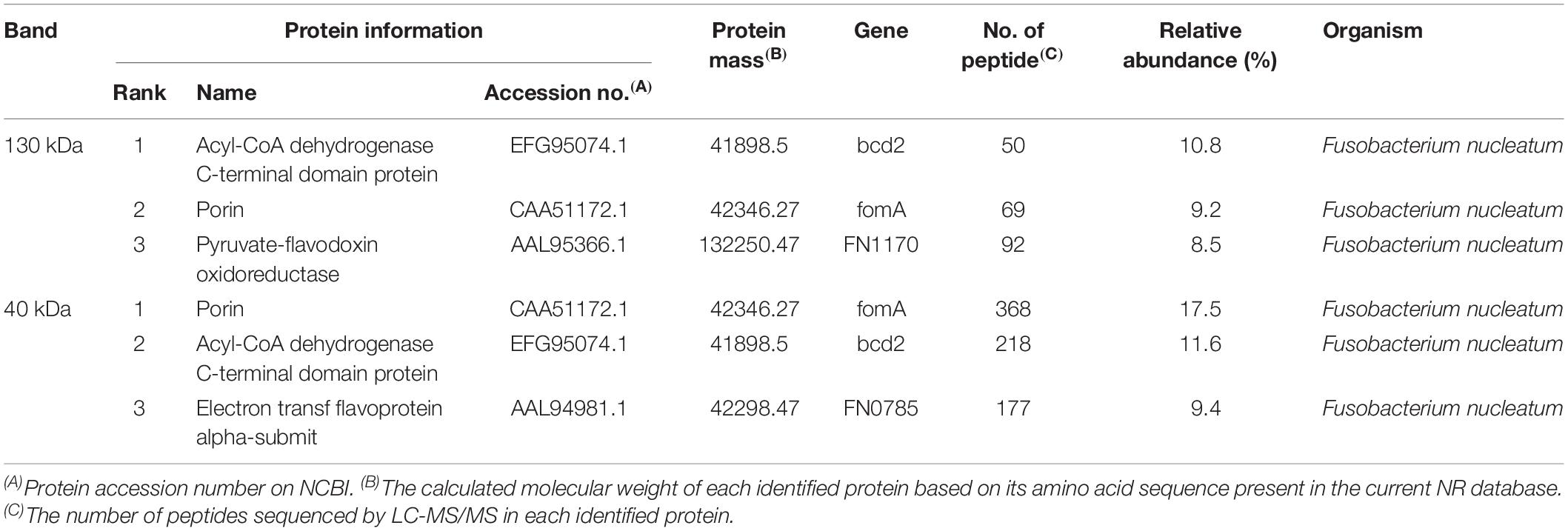
A proteomic analysis of F. nucleatum was performed by combining the protein molecular weight and functional category annotated in eggNOG. By filtering the results, 57 proteins in the M category (cell wall/membrane/envelope biogenesis) (Singer and Nicolson, 1972; Hedin et al., 2011) with an approximate molecular mass of 40 kDa and none at 130 kDa were filtered out. Next, the two strongly reactive bands at 40 and 130 kDa were cut out of the gels and sent for mass spectrometry. The sequence coverage of all the proteins identified by mass spectrometry is shown in the Supplementary Table 4. Also, six major proteins were identified. Of these, three belonged to the band at 40 kDa and the other three belonged to that at 130 kDa (Table 2), which were identified by NanoLC-ESI-MS/MS. Among these proteins, FomA, which is a major outer membrane protein of F. nucleatum, appeared in both and belonged to the proteins in the M category selected above. Thus, FomA was identified as a possible key protein eliciting the immune response.
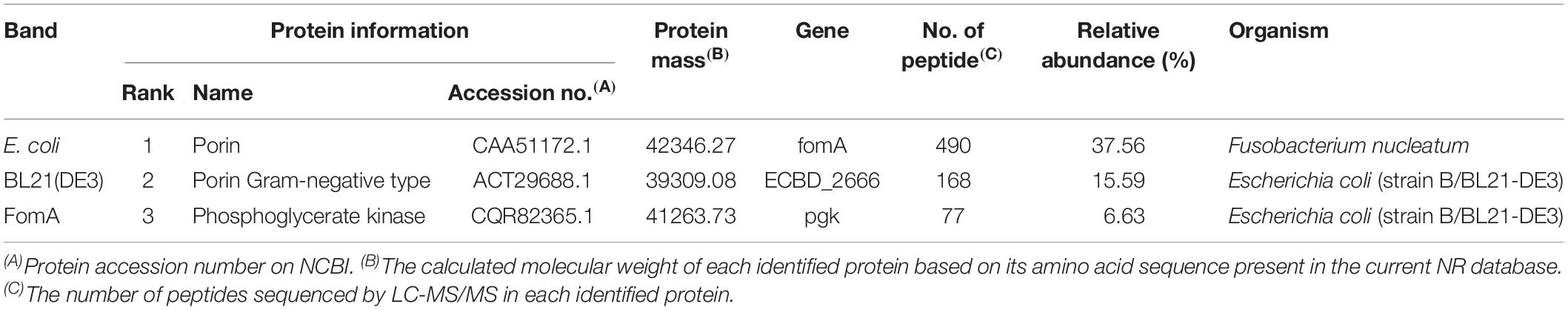
To test this hypothesis, the fomA gene was recombined into an E. coli strain harboring the expression vector. A strong reactive band with a molecular mass of 40 kDa was observed in the western blotting test using the recombinant FomA and the F. nucleatum-reactive FSN, whereas no band was observed at 40 kDa using the FomA-negative E. coli (Figure 7H). The strong band at 40 kDa was then cut and sent for mass spectrometry, the results of which further confirmed that FomA was expressed at 40 kDa of recombinant E. coli proteins (Table 3). Other details of mass spectrometry are shown in the Supplementary Table 4. Thus, these results collectively suggested that FomA was the antigen recognized by the symptom-associated antibody.
Discussion
According to our findings, the genus Fusobacterium remarkably increased in IBS-D. F. nucleatum could cause intestinal microbial dysbiosis and exacerbate visceral hypersensitivity in MS rats. The oral administration of F. nucleatum activated the secretion of specific secretory IgA that acted against it, while F. nucleatum-specific IgA was also detected in the FSN of IBS-D patients. FomA was further identified as the target antigen that bound to the F. nucleatum-specific IgA.
Microbial dysbiosis is closely associated with the occurrence and development of IBS (Ghoshal et al., 2012; Bennet et al., 2015). Some characteristics, such as an increased ratio of Firmicutes to Bacteroides and decreased bacterial diversity, have been observed in IBS patients (Simren et al., 2013; Jalanka-Tuovinen et al., 2014). However, there are still controversies regarding the characteristic bacteria in the intestinal microbiota of IBS patients. In this study, it was first revealed that the abundance of Fusobacterium was observed to be significantly higher and be positively correlated with SDS in IBS-D patients. Therefore, Fusobacterium was shown to be involved in the pathophysiology of IBS, especially with respect to mental and sensory abnormalities.
Because Fusobacterium was obviously enriched in IBS-D, F. nucleatum was chosen to administer to rats. Both MS and gavage-introduced F. nucleatum were observed to reduce microbial diversity. The results from the NMDS analysis also indicated the separation between the control and the treatment rats. In addition, gavage-introduced F. nucleatum was observed to exacerbate the visceral hypersensitivity of MS rats. Visceral hypersensitivity is considered to be an important feature of IBS (Farzaei et al., 2016; Simren et al., 2017). However, the relationship between intestinal microbiota and visceral hypersensitivity still remains unclear. As a member of the genus Fusobacterium, F. nucleatum has been demonstrated to promote the release of intestinal inflammatory factors and destroy the intestinal barrier function (Liu et al., 2007; Han, 2015; Tang et al., 2016) and is associated with oral pain and cold sensitivity (Hahn et al., 1993; Massey et al., 1993). After the gavage of F. nucleatum, the decrease in the genus Lactobacillus was detected. Lactobacillus is a type of probiotic that is used to regulate the intestinal microbiota of IBS (Niedzielin et al., 2001; Ducrotte et al., 2012). Probiotic treatment with Lactobacillus was suggested to prevent visceral hypersensitivity to colonic inflammation and an acute psychological stress (Darbaky et al., 2017). The phylum Firmicutes and the class Clostridia were also shown be present at higher levels after the gavage of F. nucleatum, and an increase in Firmicutes and some Clostridium spp. has been noted in IBS patients (Matto et al., 2005; Krogius-Kurikka et al., 2009). Therefore, the intestinal microbial dysbiosis caused by F. nucleatum might be related to visceral hypersensitivity.
Although F. nucleatum supposedly caused intestinal microbiota dysbiosis and exacerbated visceral hypersensitivity in rats, the strain was not detected in fecal samples of rats by 16s rRNA sequencing or PCR. To identify the mechanism of the colonization-independent process, the specific IgA against F. nucleatum in the FSN was evaluated. At weeks 8 and 12, the levels of F. nucleatum-specific IgA in FSN of rats increased; similarly, that in the FSN of IBS-D patients was also increased. Moreover, the levels of F. nucleatum-specific IgA in the FSN of the IBS-D patients and HC were found to be positively correlated with the SAS and SDS. When microorganisms enter the intestine, mucosal immunity is shown to be activated by microbial antigens and induce secretory IgA release for neutralization (Macpherson and Uhr, 2004; Konrad et al., 2006). Although F. nucleatum was not colonized in rats, its specific IgA was observed to be a crucial factor influencing the bacterial community by RDA analysis. These results suggest that the production of F. nucleatum-specific IgA may play a role in the pathogenesis of IBS. Moreover, FomA (Nakagaki et al., 2010), encoded by the fomA gene (Toussi et al., 2012), was identified as the molecular antigen stimulating the production of specific IgA in the intestine. FomA is a major outer membrane protein of F. nucleatum (Nakagaki et al., 2010). Membrane proteins have been shown to play crucial roles in bioenergetics, transport, and signaling (Singer and Nicolson, 1972; Hedin et al., 2011), and more than half of the known drugs target membrane proteins (Sojo et al., 2016). Furthermore, FomA is a Toll-like receptor 2 agonist with an adjuvant immune activity that stimulates the production of IgA and IgG (Toussi et al., 2012; Ma et al., 2013), while it is also capable of recruiting oral bacteria (Liu et al., 2010). Interestingly, the oral administration of recombinant FomA to mice was shown to increase IgG in serum and IgA in saliva and reduce the risk of periodontal infection of F. nucleatum (Ma et al., 2013). Therefore, these results suggest that F. nucleatum can activate intestinal immunity via FomA to promote the secretion of high quantities of IgA that prevent its colonization.
Despite the useful findings, there are some limitations to this study. First, whether or not other strains of the Fusobacterium would have a similar effect was not determined. Second, although F. nucleatum was observed to be capable of inducing the secretion of IgA, its specific immune mechanism was not explored and requires further research.
In conclusion, Fusobacterium was observed to be an increased genus with significantly higher abundance in IBS-D patients. Moreover, the mechanism of F. nucleatum in the pathogenesis of IBS through causing microbial dysbiosis and exacerbating visceral hypersensitivity in a colonization-independent manner was confirmed. Furthermore, FomA was identified to be the antigen stimulating the production of the symptom-associated antibody.
Data Availability Statement
The datasets generated for this study can be found in the National Center for Biotechnology Information (NCBI) database with accession code PRJNA511737, National Center for Biotechnology Information (NCBI) database with accession code PRJNA511738.
Ethics Statement
The studies involving human participants were reviewed and approved by the ethics committees in Qilu Hospital, Shandong University. The patients/participants provided their written informed consent to participate in this study. The animal study was reviewed and approved by Ethical and Institutional Animal Care and Use Committee of Qilu Hospital, Shandong University.
Author Contributions
XG, ML, and XZ designed the project. ML conducted the bioinformatic data mining. XG, LS, TL, and MZ conducted key experiments. XG and ML performed statistical analysis. LL, ZL, and PW provided vital expertise and advice. XG and LS wrote the manuscript. All authors read the manuscript and approved the final draft that was submitted.
Funding
This work was supported by the National Natural Science Foundation of China (Nos. 81770538 and 81700457) and Key Research and Development Program of Shandong Province (2017CXGC1215).
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
We thank all the IBS-D patients and healthy volunteers for participating in this study and providing samples. We thank Xin Long, Lin Lin, Bing Li, and Kairuo Wang for their valuable support and advices during the research and manuscript drafting. We also thank the colleagues of Shanghai Majorbio for their assistance in microbial sequencing and analysis.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmicb.2020.01281/full#supplementary-material
References
Aagaard, K., Riehle, K., Ma, J., Segata, N., Mistretta, T. A., Coarfa, C., et al. (2012). A metagenomic approach to characterization of the vaginal microbiome signature in pregnancy. PLoS One 7:e36466. doi: 10.1371/journal.pone.0036466
Al-Chaer, E. D., Kawasaki, M., and Pasricha, P. J. (2000). A new model of chronic visceral hypersensitivity in adult rats induced by colon irritation during postnatal development. Gastroenterology 119, 1276–1285. doi: 10.1053/gast.2000.19576
Bengtson, M. B., Aamodt, G., Vatn, M. H., and Harris, J. R. (2015). Co-occurrence of IBS and symptoms of anxiety or depression, among Norwegian twins, is influenced by both heredity and intrauterine growth. BMC Gastroenterol. 15:9. doi: 10.1186/s12876-015-0237-y
Bennet, S. M., Ohman, L., and Simren, M. (2015). Gut microbiota as potential orchestrators of irritable bowel syndrome. Gut Liver 9, 318–331. doi: 10.5009/gnl14344
Crouzet, L., Gaultier, E., Del’Homme, C., Cartier, C., Delmas, E., Dapoigny, M., et al. (2013). The hypersensitivity to colonic distension of IBS patients can be transferred to rats through their fecal microbiota. Neurogastroenterol. Motil. 25, e272–e282. doi: 10.1111/nmo.12103
Darbaky, Y., Evrard, B., Patrier, S., Falenta, J., Garcin, S., Tridon, A., et al. (2017). Oral probiotic treatment of Lactobacillus rhamnosus Lcr35((R)) prevents visceral hypersensitivity to a colonic inflammation and an acute psychological stress. J. Appl. Microbiol. 122, 188–200. doi: 10.1111/jam.13320
De Palma, G., Lynch, M. D., Lu, J., Dang, V. T., Deng, Y., Jury, J., et al. (2017). Transplantation of fecal microbiota from patients with irritable bowel syndrome alters gut function and behavior in recipient mice. Sci. Transl. Med. 9:aaf6397. doi: 10.1126/scitranslmed.aaf6397
Donaldson, G. P., Ladinsky, M. S., Yu, K. B., Sanders, J. G., Yoo, B. B., Chou, W. C., et al. (2018). Gut microbiota utilize immunoglobulin A for mucosal colonization. Science 360, 795–800. doi: 10.1126/science.aaq0926
Ducrotte, P., Sawant, P., and Jayanthi, V. (2012). Clinical trial: Lactobacillus plantarum 299v (DSM 9843) improves symptoms of irritable bowel syndrome. World J. Gastroenterol. 18, 4012–4018. doi: 10.3748/wjg.v18.i30.4012
Enck, P., Aziz, Q., Barbara, G., Farmer, A. D., Fukudo, S., Mayer, E. A., et al. (2016). Irritable bowel syndrome. Nat. Rev. Dis. Primers 2:16014. doi: 10.1038/nrdp.2016.14
Farzaei, M. H., Bahramsoltani, R., Abdollahi, M., and Rahimi, R. (2016). The role of visceral hypersensitivity in irritable bowel syndrome: pharmacological targets and novel treatments. J. Neurogastroenterol. Motil. 22, 558–574. doi: 10.5056/jnm16001
Francis, C. Y., Morris, J., and Whorwell, P. J. (1997). The irritable bowel severity scoring system: a simple method of monitoring irritable bowel syndrome and its progress. Aliment Pharmacol. Ther. 11, 395–402. doi: 10.1046/j.1365-2036.1997.142318000.x
Ghoshal, U. C., Shukla, R., Ghoshal, U., Gwee, K. A., Ng, S. C., and Quigley, E. M. (2012). The gut microbiota and irritable bowel syndrome: friend or foe? Int. J. Inflam. 2012:151085. doi: 10.1155/2012/151085
Greenwood-Van Meerveld, B., and Johnson, A. C. (2017). Stress-induced chronic visceral pain of gastrointestinal origin. Front. Syst. Neurosci. 11:86. doi: 10.3389/fnsys.2017.00086
Griffin, N. M., Yu, J., Long, F., Oh, P., Shore, S., Li, Y., et al. (2010). Label-free, normalized quantification of complex mass spectrometry data for proteomic analysis. Nat. Biotechnol. 28, 83–89. doi: 10.1038/nbt.1592
Hahn, C. L., Falkler, W. A., and Minah, G. E. (1993). Correlation between thermal sensitivity and microorganisms isolated from deep carious dentin. J. Endod. 19, 26–30. doi: 10.1016/s0099-2399(06)81037-x
Han, Y. W. (2015). Fusobacterium nucleatum: a commensal-turned pathogen. Curr. Opin. Microbiol. 23, 141–147. doi: 10.1016/j.mib.2014.11.013
Hedin, L. E., Illergard, K., and Elofsson, A. (2011). An introduction to membrane proteins. J. Proteome Res. 10, 3324–3331. doi: 10.1021/pr200145a
Jalanka-Tuovinen, J., Salojarvi, J., Salonen, A., Immonen, O., Garsed, K., Kelly, F. M., et al. (2014). Faecal microbiota composition and host-microbe cross-talk following gastroenteritis and in postinfectious irritable bowel syndrome. Gut 63, 1737–1745. doi: 10.1136/gutjnl-2013-305994
Konrad, A., Cong, Y., Duck, W., Borlaza, R., and Elson, C. O. (2006). Tight mucosal compartmentation of the murine immune response to antigens of the enteric microbiota. Gastroenterology 130, 2050–2059. doi: 10.1053/j.gastro.2006.02.055
Krogius-Kurikka, L., Lyra, A., Malinen, E., Aarnikunnas, J., Tuimala, J., Paulin, L., et al. (2009). Microbial community analysis reveals high level phylogenetic alterations in the overall gastrointestinal microbiota of diarrhoea-predominant irritable bowel syndrome sufferers. BMC Gastroenterol. 9:95. doi: 10.1186/1471-230x-9-95
Liu, H., Redline, R. W., and Han, Y. W. (2007). Fusobacterium nucleatum induces fetal death in mice via stimulation of TLR4-mediated placental inflammatory response. J. Immunol. 179, 2501–2508. doi: 10.4049/jimmunol.179.4.2501
Liu, P. F., Shi, W., Zhu, W., Smith, J. W., Hsieh, S. L., Gallo, R. L., et al. (2010). Vaccination targeting surface FomA of Fusobacterium nucleatum against bacterial co-aggregation: implication for treatment of periodontal infection and halitosis. Vaccine 28, 3496–3505. doi: 10.1016/j.vaccine.2010.02.047
Lovell, R. M., and Ford, A. C. (2012). Global prevalence of and risk factors for irritable bowel syndrome: a meta-analysis. Clin. Gastroenterol. Hepatol. 10, 712–721. doi: 10.1016/j.cgh.2012.02.029
Ma, L., Ding, Q., Feng, X., and Li, F. (2013). The protective effect of recombinant FomA-expressing Lactobacillus acidophilus against periodontal infection. Inflammation 36, 1160–1170. doi: 10.1007/s10753-013-9651-x
Macpherson, A. J., and Uhr, T. (2004). Induction of protective IgA by intestinal dendritic cells carrying commensal bacteria. Science 303, 1662–1665. doi: 10.1126/science.1091334
Malinen, E., Krogius-Kurikka, L., Lyra, A., Nikkila, J., Jaaskelainen, A., Rinttila, T., et al. (2010). Association of symptoms with gastrointestinal microbiota in irritable bowel syndrome. World J. Gastroenterol. 16, 4532–4540.
Massey, W. L., Romberg, D. M., Hunter, N., and Hume, W. R. (1993). The association of carious dentin microflora with tissue changes in human pulpitis. Oral Microbiol. Immunol. 8, 30–35. doi: 10.1111/j.1399-302x.1993.tb00539.x
Matto, J., Maunuksela, L., Kajander, K., Palva, A., Korpela, R., Kassinen, A., et al. (2005). Composition and temporal stability of gastrointestinal microbiota in irritable bowel syndrome–a longitudinal study in IBS and control subjects. FEMS Immunol. Med. Microbiol. 43, 213–222. doi: 10.1016/j.femsim.2004.08.009
Nakagaki, H., Sekine, S., Terao, Y., Toe, M., Tanaka, M., Ito, H. O., et al. (2010). Fusobacterium nucleatum envelope protein FomA is immunogenic and binds to the salivary statherin-derived peptide. Infect. Immun. 78, 1185–1192. doi: 10.1128/iai.01224-09
Niedzielin, K., Kordecki, H., and Birkenfeld, B. (2001). A controlled, double-blind, randomized study on the efficacy of Lactobacillus plantarum 299V in patients with irritable bowel syndrome. Eur. J. Gastroenterol. Hepatol. 13, 1143–1147. doi: 10.1097/00042737-200110000-00004
O’Mahony, S. M., Hyland, N. P., Dinan, T. G., and Cryan, J. F. (2011). Maternal separation as a model of brain-gut axis dysfunction. Psychopharmacology 214, 71–88. doi: 10.1007/s00213-010-2010-9
Qin, J., and Zhang, X. (2002). Identification of in vivo protein phosphorylation sites with mass spectrometry. Methods Mol. Biol. 194, 211–221. doi: 10.1385/1-59259-181-7:211
Sandrini, S., Aldriwesh, M., Alruways, M., and Freestone, P. (2015). Microbial endocrinology: host-bacteria communication within the gut microbiome. J. Endocrinol. 225, R21–R34. doi: 10.1530/joe-14-0615
Schloss, P. D., Westcott, S. L., Ryabin, T., Hall, J. R., Hartmann, M., Hollister, E. B., et al. (2009). Introducing mothur: open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl. Environ. Microbiol. 75, 7537–7541. doi: 10.1128/aem.01541-09
Segata, N., Haake, S. K., Mannon, P., Lemon, K. P., Waldron, L., Gevers, D., et al. (2012). Composition of the adult digestive tract bacterial microbiome based on seven mouth surfaces, tonsils, throat and stool samples. Genome Biol. 13:R42. doi: 10.1186/gb-2012-13-6-r42
Simren, M., Barbara, G., Flint, H. J., Spiegel, B. M., Spiller, R. C., Vanner, S., et al. (2013). Intestinal microbiota in functional bowel disorders: a Rome foundation report. Gut 62, 159–176. doi: 10.1136/gutjnl-2012-302167
Simren, M., Tornblom, H., Palsson, O. S., van Tilburg, M. A., Van Oudenhove, L., Tack, J., et al. (2017). Visceral hypersensitivity is associated with GI symptom severity in functional GI disorders: consistent findings from five different patient cohorts. Gut 67, 255–262. doi: 10.1136/gutjnl-2016-312361
Simren, M., Tornblom, H., Palsson, O. S., van Tilburg, M. A. L., Van Oudenhove, L., Tack, J., et al. (2018). Visceral hypersensitivity is associated with GI symptom severity in functional GI disorders: consistent findings from five different patient cohorts. Gut 67, 255–262. doi: 10.1136/gutjnl-2016-312361
Singer, S. J., and Nicolson, G. L. (1972). The fluid mosaic model of the structure of cell membranes. Science 175, 720–731. doi: 10.1126/science.175.4023.720
Sojo, V., Dessimoz, C., Pomiankowski, A., and Lane, N. (2016). Membrane proteins are dramatically less conserved than water-soluble proteins across the tree of life. Mol. Biol. Evol. 33, 2874–2884. doi: 10.1093/molbev/msw164
Tang, B., Wang, K., Jia, Y. P., Zhu, P., Fang, Y., Zhang, Z. J., et al. (2016). Fusobacterium nucleatum-induced impairment of autophagic flux enhances the expression of proinflammatory cytokines via ROS in Caco-2 cells. PLoS One 11:e0165701. doi: 10.1371/journal.pone.0165701
Tang, R., Wei, Y., Li, Y., Chen, W., Chen, H., Wang, Q., et al. (2018). Gut microbial profile is altered in primary Biliary cholangitis and partially restored after UDCA therapy. Gut 67, 534–541. doi: 10.1136/gutjnl-2016-313332
Toussi, D. N., Liu, X., and Massari, P. (2012). The FomA porin from Fusobacterium nucleatum is a Toll-like receptor 2 agonist with immune adjuvant activity. Clin. Vaccine Immunol. 19, 1093–1101. doi: 10.1128/cvi.00236-12
Wang, H. F., Li, L. F., Guo, S. H., Zeng, Q. Y., Ning, F., Liu, W. L., et al. (2016). Evaluation of antibody level against Fusobacterium nucleatum in the serological diagnosis of colorectal cancer. Sci. Rep. 6:33440. doi: 10.1038/srep33440
Wang, P., Du, C., Chen, F. X., Li, C. Q., Yu, Y. B., Han, T., et al. (2016). BDNF contributes to IBS-like colonic hypersensitivity via activating the enteroglia-nerve unit. Sci. Rep. 6:20320. doi: 10.1038/srep20320
Xu, D., Wu, X., Grabauskas, G., and Owyang, C. (2013). Butyrate-induced colonic hypersensitivity is mediated by mitogen-activated protein kinase activation in rat dorsal root ganglia. Gut 62, 1466–1474. doi: 10.1136/gutjnl-2012-302260
Zhao, Q., Ji, M., Li, R., and Ren, Z. J. (2017). Long-term performance of sediment microbial fuel cells with multiple anodes. Bioresour. Technol. 237, 178–185. doi: 10.1016/j.biortech.2017.03.002
Zhou, X. Y., Li, M., Li, X., Long, X., Zuo, X. L., Hou, X. H., et al. (2016). Visceral hypersensitive rats share common dysbiosis features with irritable bowel syndrome patients. World J. Gastroenterol. 22, 5211–5227. doi: 10.3748/wjg.v22.i22.5211
Keywords: Fusobacterium nucleatum, irritable bowel syndrome, visceral hypersensitivity, intestinal microbiota, immunoglobulin A
Citation: Gu X, Song L, Li L, Liu T, Zhang M, Li Z, Wang P, Li M and Zuo X (2020) Fusobacterium nucleatum Causes Microbial Dysbiosis and Exacerbates Visceral Hypersensitivity in a Colonization-Independent Manner. Front. Microbiol. 11:1281. doi: 10.3389/fmicb.2020.01281
Received: 15 December 2019; Accepted: 19 May 2020;
Published: 24 June 2020.
Edited by:
Esaki M. Shankar, Central University of Tamil Nadu, IndiaReviewed by:
Huaxi Yi, Ocean University of China, ChinaGislane Lelis Vilela de Oliveira, São Paulo State University, Brazil
Copyright © 2020 Gu, Song, Li, Liu, Zhang, Li, Wang, Li and Zuo. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Xiu-li Zuo, enVveGl1bGlAc2R1LmVkdS5jbg==; Ming Li, bGltaW5nZWNob0AxMjYuY29t
 Xiang Gu1,2
Xiang Gu1,2 Li-jin Song
Li-jin Song Li-xiang Li
Li-xiang Li Ming Li
Ming Li Xiu-li Zuo
Xiu-li Zuo