- 1Institute of Hematology, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China
- 2Department of Integrated Traditional Chinese and Western Medicine, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China
- 3Institute for Cardiovascular Prevention, Ludwig-Maximilians-University, Munich, Germany
Tertiary lymphoid organs (TLOs) frequently develop locally in adults in response to non-resolving inflammation. Chronic inflammation leads to the differentiation of stromal fibroblast cells toward lymphoid tissue organizer-like cells, which interact with lymphotoxin immune cells. The interaction initiates lymphoid neogenesis by recruiting immune cells to the site of inflammation and ultimately leads to the formation of TLOs. Mature TLOs harbor a segregated T-cell zone, B-cell follicles with an activated germinal center, follicular dendritic cells, and high endothelial venules, which architecturally resemble those in secondary lymphoid organs. Since CXCL13 and LTα1β2 play key roles in TLO neogenesis, they might constitute potential biomarkers of TLO activity. The well-developed TLOs actively regulate local immune responses and influence disease progression, and they are thereby regarded as the powerhouses of local immunity. In this review, we recapitulated the determinants for TLOs development, with great emphasis on the fundamental role of chronic inflammation and tissue-resident stromal cells for TLO neogenesis, hence offering guidance for therapeutic interventions in TLO-associated diseases.
Introduction
Inflammation is a process of physiological responses to pathological stimuli that involves immune cells and tissue-resident cells. The immune cells are mobilized to migrate to the infected or damaged tissues through the vasculature, and they release inflammatory mediators, such as cytokines and prostaglandins. These cells and inflammatory mediators are important players within the inflammatory microenvironment, although they differ at different phases of inflammation. At the early stage, the acute inflammatory response takes place within the first few hours due mainly to the infiltration of innate immune cells, including neutrophils, and is typically characterized by the appearance of redness, swollenness, and pain symptoms (1). At the later stage, the chronic cellular response involves the recruitment of not only innate immune cells but also adaptive immune cells. When antigens cannot be removed, the inflammation persists as chronic non-resolving inflammation in a site-specific manner. Such chronic inflammation serves as a common basis for immune cell-tissue resident stromal cell interactions and the accumulation of immune cells at sites of inflammation in a plethora of clinical diseases. The aggregates form segregated T-cell areas and B-cell follicles with follicular dendritic cells (FDCs) as well as neovascularization with newly formed blood vessels, lymph vessels, and high endothelial venules (HEVs) (2, 3). These structures have been described as tertiary lymphoid organs (TLOs), tertiary lymphoid structures, ectopic lymphoid organs, and lymphoid tissue neogenesis (4–6).
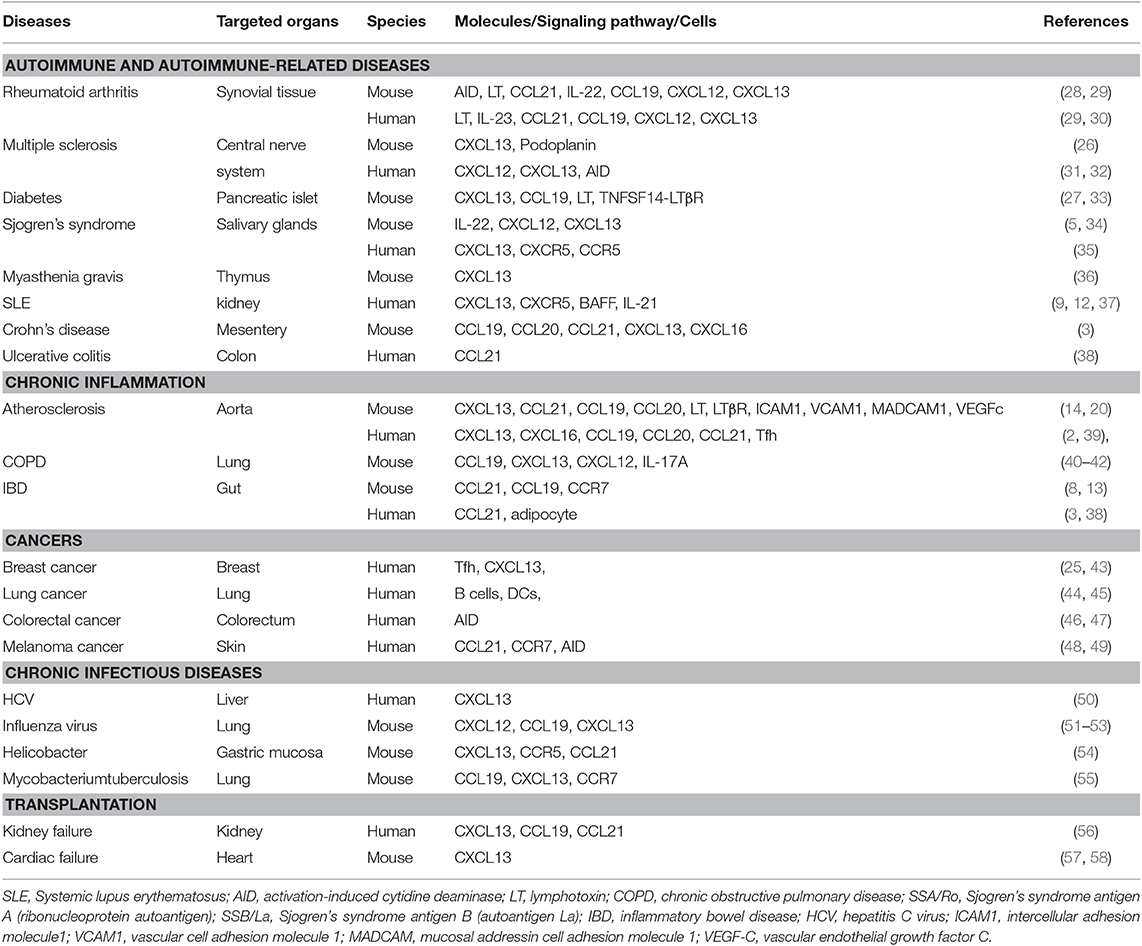
TLOs develop as ectopic lymphocyte clusters in connective tissues or parenchyma of diseased organs in various non-resolving chronic inflammatory diseases, including autoimmune diseases (7, 8), cancers (9), chronic infectious diseases (10), transplant rejections (11), and chronic inflammatory diseases like atherosclerosis (6, 12) and inflammatory bowel disease (13), indicating that chronic inflammation is the common promoter of TLO neogenesis. TLOs share remarkable cellular and structural similarities with secondary lymphoid organs (SLOs); for example, both contain various innate and adaptive immune cell types, T-cell areas, B-cell follicles, and HEVs (14). Both TLOs and SLOs require similar cellular and molecular developmental signals, albeit of their different source of origins. TLOs may play significant roles in local primary immune responses by mimicking SLO functions and, to some extent, can serve as protective or detrimental to organisms, though they are not encapsulated and can occur in any diseased tissues, unlike SLOs.
Cumulative evidence indicates that the ectopic expression of homeostatic lymphoid chemokines (i.e., CXCL13, CCL19, and CCL21) plays a vital role for TLO formation and contributes to leukocyte recruitment and their persistence in the tissues, while deficiency in any of these molecules can abrogate TLO formation. The resident stromal cells, including fibroblasts, endothelial cells, pericytes, and vascular smooth muscle cells (VSMCs), are the original producers of the chemokines that can functionally influence the ectopic infiltration of immune cells at the site of chronic inflammation (15, 16). Since TLOs harbor a number of immune cells, they may initiate local immune responses and ultimately influence the progression of diseases. In this review, we have summarized the current knowledge of TLOs—their definition, location, molecular determinants, and the potential roles they have in diseases—and discuss the functions of chronic inflammation and stromal cells in TLO formation.
TLO Development
The Definition of a TLO
TLOs are immune cell aggregates within or adjacent to the local tissues associated with chronic inflammation. TLOs share a structural homology with SLOs, including T-cell zones, B-cell follicles, stromal cells, HEV, blood, and lymphoid vessels. TLOs develop postnatally at undefined locations and are significantly associated with overwhelming antigen stimuli. SLO and primary lymphoid organ development, meanwhile, always occur in highly coordinated processes at predetermined locations during embryogenesis (17). The anatomy of TLOs is quite plastic, and the cellular composition varies from one model to the other. However, T/B cell segregation, vascular specialization, and certain lymphorganogenic chemokine expressions are the basic characteristics of TLOs; the well-developed TLOs with a germinal center (GC) and HEV formation are highly dependent on the disease stage and on the efficiency and duration of antigen-driven responses at the local site. Though TLOs have been extensively studied in the last 10 years, the standard definition of TLOs has not yet been set. Recently, Neyt et al. (18) proposed six criteria for determining TLOs: the presence of organized infiltrated T and B cells; the appearance of T-cell areas containing fibroblast reticular cell (FRC) networks; the presence of HEV; B cell class switching, GC reaction; activation-induced cytidine deaminase (AID) enzyme; and FDC presentation. However, it is not feasible to apply all of these criteria to each disease model nor is it possible to meet all criteria in different developing stages of the same disease. Based on the studies performed on artery TLO (ATLO) (13, 19, 20), we have suggested a definition of an ATLO based on three developmental stages: stage I has mixed T- and B-cell infiltrates; stage II contains segregated T- and B-cell areas with lymph vessels, HEVs, and conduits; and stage III has well-structured TLOs with segregated T- and B-cell follicles, activated GCs, and FDCs (Figure 1). However, these “criteria” are based on specific disease models and may be not applicable to other disease models as murine TLOs might differ from those in humans. Therefore, great effort is needed to set up the common criteria for defining TLOs for all disease models.

Figure 1. TLO stage classification. Stage 0, normal tissue without TLO formation; Stage I, early TLOs with mixed T/B-cell aggregates; Stage II, pre-TLOs with segregated T/B-cell area with lymph vessels and conduits; Stage III, well organized TLOs containing segregated T-cell area and B-cell follicles with germinal centers and follicle DC network.
The Spontaneous- and Induced- TLOs
TLOs can spontaneously develop in tissues with non-resolving chronic inflammation caused by persistent inflammatory stimuli or self-antigen during the disease progression. Their locations therefore vary depending on the disease models; tumor-associated TLOs, for example, are located peritumorally (21–23), intratumorally (24), or mixed (25). However, ATLOs are usually found in the adventitia of aged chow-fed Apoe−/− mice (20) (Figure 2), which start to develop at around 55 weeks old post-birth and are fully formed at around 78 weeks old. Moreover, ATLO size correlates with the severity of intima atherosclerosis, and TLO-like structures were absent in the non-atherosclerotic arterial wall, thus indicating the importance of the local inflammation in the initiation and progression of ATLOs. Also, TLOs could develop in the central nervous system in autoimmune diseases, such as in the brain in multiple sclerosis (26). In the genetic non-obese diabetic (NOD) mouse model, TLOs arose in the pancreas with increased size and highly organized structure during disease progression (27). The locations of TLOs are shown in Table 1. In addition, TLOs can also be induced experimentally in some murine models. Administrating pathogens or inflammatory substances and the overexpression of lymphocyte-recruiting chemokines can result in the organoid assembly of lymphocytes (59). For example, significant lymphocytic infiltrates were detected in the thyroid gland of mice with CCL21 overexpression (60), and the inducible bronchus-associated lymphoid tissue (iBALT) developed in the lung of mice injected with a lipopolysaccharide (LPS) or influenza infection (51–53). The major difference between spontaneous and induced TLOs is that strong external stimuli are needed for the later for the induction of TLO formation, suggesting that there are difference in the intensities of immune responses. The immune activity in induced TLOs is variable, depending on the intensity of the stimuli applied to trigger them compared to spontaneous TLOs where immune activity might be low due to the prolonged chronic stimulation. However, the distinction between spontaneous and induced TLOs according to their immune activity and persistence is ambiguous since there is no standard to grade immune activity.
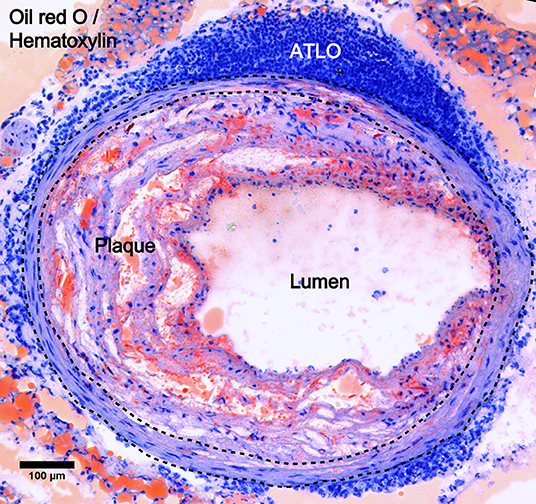
Figure 2. ATLO location in atherosclerotic aorta. Oil red O/Hematoxylin staining showing ATLO position in the abdominal aorta adventitia relative to media (dashed lines) and intimal plaque in aged Apoe−/− mice.
Chronic Inflammation Favors Lymphoid Neogenesis
Inflammation is a self-limiting process, and multiple mechanisms ensure resolution in the normal condition. It is also a basic mediator of many diseases. TLOs are more frequently formed in chronic inflammatory diseases, although they are not present in all chronic inflammation. In contrast to SLO development, the inflammatory microenvironment provides the initial signal for TLO neogenesis, and chronic inflammation is sufficient to induce TLO formation even without the presence of lymphoid tissue inducer (LTi) cells (61), indicating that chronic inflammation is a fundamental player that favors lymphoid neogenesis.
Under chronic inflammatory conditions, resident stromal mesenchymal cells (62), such as fibroblasts, endothelial cells, VSMCs, and pericytes, are activated by proinflammatory molecules, including TNFα, LTα1β2, IL-17, IL-23, and chemokines, including CCL2, CCL3, CXCL9, CXCL10, and CXCL11 (1, 6, 63, 64). After activation, these cells are transformed into lymphoid tissue organizer (TLo)-like cells to release lymphorganogenic chemokines, such as CXCL13 and CCL21, to initiate the formation of TLOs (43, 62). For example, TNFα and LTα1β2, by interacting with TNFR1 and LTβR, respectively, activate medial VSMCs to acquire the phenotype of LTo cells. Most importantly, these activated VSMCs themselves produce high levels of CXCL13 and CCL21, thus promoting TLO formation in adventitia in atherosclerosis (14, 65). Moreover, the upregulated integrins on LTo or LTo-like cells, such as vascular cell adhesion molecule-1 and intercellular cell adhesion molecule-1, also facilitate the recruitment of immune cells to the local chronic inflammatory site, thus contributing to TLO formation (14, 15).
Locally or systemically, chronic inflammation persistently activates immune cells, upregulates LTα1β2 expression on their cell surfaces and releases cytokines, chemokines, or tissue factors, which act as indirect regulators of lymphoid organogenesis (66). Overexpression of TNFα by myeloid cells could induce intestinal TLOs in the absence of LTi cells (61). Similarly, IL-17A produced by Th17 cells alone overcomes the absence of LTi cells and mediates the formation of iBALT. The increased LTα-independent expression of CXCL13 was important for follicle formation during iBALT neogenesis (4). Both Th17 and LTi cells produce IL-22. Interestingly, IL-22−/− mice showed a significant defect in early TLO formation and chemokine secretion because IL-22 is necessary for CXCL13 expression in fibroblast cells (5). In conclusion, regardless of the sources of the environmental mediators, the persistence of an inflammatory microenvironment is a clear factor that favors TLO formation.
Roles of Innate and Adaptive Immune Cells in TLOs
Circulating monocyte-derived macrophages are known to migrate to certain tissues upon chronic inflammation, such as the salivary gland in Sjogren's syndrome (SS), and become a source of chemokines, such as CXCL13 and CXCL12, to promote TLO development (67). Unlike classical macrophages, tissue-resident macrophages are believed to be maintained by local proliferation and high expression of IL-7Rα (68), which is similar to LTi cells. It was reported that IL-7, together with CXCR5, promoted IL-7R upregulation in lymphoid organs (69). Moreover, overexpression of IL-7 resulted in TLO formation peripherally in a lymphotoxin (LT)α-dependent manner (70). It is possible that tissue-resident macrophages act as LTi cells and interact with IL-7-expressing stromal fibroblasts to initiate TLO formation. Moreover, TNF-expressing myeloid cells, such as macrophage and inflammatory monocytes, were required for fat-associated lymphoid clusters following in inflammatory challenge (71), and CD11c+ DCs were necessary for the maintenance of TLOs in the LPS-induced iBALT mouse model (72).
B cells are the predominant cell population during TLO neogenesis. In the later stage, advanced TLOs display features of B cell follicles comprising active GCs, indicating the proliferation and differentiation of reactive B cells. They could not only generate antibodies for immune responses, but also support local immune cell activation within the inflammatory tissues (6), and they play a critical role in both the initiation and organization of TLOs. B cells are important candidates for LT production, indicating that they might have a potential role in TLO neogenesis (73). Gene expression studies showed that B cells are recruited into TLOs by the interaction of chemokines CXCL13 and CXCL10 with their receptors CXCR5 and CXCR3, respectively (74, 75). Depletion of B cells from synovial tissue led to a reduction in T-cell derived IFNγ and IL-10 production, suggesting that synovial B cells control T-cell responses. In a chronic inflammatory mouse model, ATLOs harbor a high frequency of B cells which secrete large amounts of IgM and IgG antibodies. Although less numerous than B2 B cells, IL-10-expressing B1b cells present in ATLOs might exert key regulatory functions (76). Whether B1 B cells contribute to TLO maturation is not clear so far. The secreted antibodies by TLO B cells may have different effects on disease progression. These antibodies may serve as a double-edged sword, which could either have a positive role in infectious diseases or exacerbate autoimmune diseases. Since TLO B cells were also found in the synovium of inflamed joints, Rituximab (an anti-CD20 B-cell depleting antibody) might be an alternative therapy for treating rheumatoid arthritis, besides of TNF-α and IL-1 antagonist in clinic. It should know that Rituximab is an antibody for B-cell therapy in general, but not TLO specific. The role of B cells in the formation of TLOs in other autoimmune diseases, transplantation, and tumorigenesis events has been extensively reported (77). Therefore, the B cell aggregates could be regarded as the “marker” for TLO formation.
Regulatory T cells (Tregs) are believed to regulate the suppressive function in TLOs (6). In aged Apoe−/− mice, both natural Tregs and induced Tregs were activated in ATLO during non-resolving chronic inflammation (20). Tregs in ATLOs may suppress the immune responses in plaque in a certain way because disruption of ATLOs by depletion of LTβR globally or specifically in VSMCs can abolish TLO formation and enhance atherosclerosis, suggesting that ATLOs have a protective role in disease progression (20). However, Clement M. et al. reported that regulatory CD8+ T cells controlled the development of TLOs and atherosclerosis, suggesting a pro-atherosclerotic role of TLOs in atherosclerosis (39). These conflicting conclusions from two individual studies might be due to some exterior factors, such as age, stage of disease, and environmental factors. Further studies are therefore warranted to investigate the precise role of TLOs in atherosclerosis.
The Molecular Determinants of TLOs
The development of SLOs is a complex process that involves the interaction of LTα1β2-expressing hematopoietic LTi cells with LTβR-expressing non-hematopoietic stromal LTo cells. This interaction leads to the release of the cascade of adhesion molecules (e.g., intercellular adhesion molecule 1 and vascular cell adhesion molecule 1), cytokines (e.g., IFNγ, IL-17, IL-22, and IL-23) chemokines (e.g., CCL19, CCL21, CXCL12, and CXCL13), and the LTβR signaling pathway (18, 62). These secreted molecules and angiogenic growth factors regulate cellular and structural development of SLOs, while lymphangiogenic growth factors, like vascular endothelial growth factor C, induce lymphatic vessel development (78). Furthermore, LTo cells transdifferentiate into HEVs, FRCs, or FDCs, forming a network and facilitating T- and B-cell migration. The studies of TLOs in last 20 years have largely expanded our understanding of the molecular mechanisms involved in TLO formation. Though TLOs greatly resemble SLOs in cellularity and structure, the molecular determinants might be different in a certain way. Here, we discussed the vital role of TNFSF members, cytokines and chemokines that contribute TLO development.
LT, LIGHT, and LTβR
The interaction of TNF superfamily members, i.e., LTα, LTβ, LIGHT (homologous to LT, also known as TNFSF14), and their receptor LTβR plays a critical role for lymphoid organ development. Over-expression of LTα alone or LTα and LTβ co-expression under rat insulin promoter II induced the development of LN-like structures with organized T- and B-cell areas and HEV in murine models (79), while LTα deficiency disorganized lymphocyte aggregation and HEV differentiation (51). In the NOD mouse model, LIGHT and LTβR expression were increased in the pancreatic TLOs (33), and the overexpression of LIGHT aggravated the disease (27). In addition, the overexpression of LTα or LIGHT induced by tumor cells resulted in intra-tumoral TLO development, indicating that the LTβR signaling pathway is crucial for the development of TLOs. Activation of LTβR in VSMCs was implicated in ATLO formation, whereas the interruption of the LTβR signaling pathway disturbed TLO formation with significantly reduced lymphocytic chemokine expression and HEVs development (20). Collectively, these data demonstrated that the LT-LTβR signaling pathway is crucial for the development, maintenance, and organization of TLOs.
Cytokines
It is becoming increasingly clear that IL-17 is an important mediator for TLO development. The administration of LPS, viruses, or M. tuberculosis infection induced iBALT formation in an IL-17-dependent manner (80). IL-17 promotes inflammatory and homeostatic chemokine production, which is critical for iBALT initiation, while LT signaling is required for the differentiation of FRCs, FDC, and HEV formation in the later step of iBALT development. Moreover, IL-17+ CD4 T cells trigger TLO neogenesis in the central nervous system in the experimental autoimmune encephalitis model (26). TCRγδ T cells-derived IL-17 trigger stromal cells to release CXCL12 and thereby induce follicle formation in iBALT even in absence of FDCs (81). These data demonstrate that IL-17 is crucial for TLO formation.
Overexpression of IL-5 in the respiratory epithelium induced the formation of organized iBALT with epithelial hypertrophy, goblet cell hyperplasia, and accumulation of eosinophils in the airway lumen (82). IL-7, together with CXCR5, promotes TLO formation, and the overexpression of IL-7 thus resulted in TLO formation in non-lymphoid tissues (70). Furthermore, IL-27 inhibits TLO development and was proposed to be a novel therapeutic target in clinical treatment (83).
TNFα is known to promote the receptor expression of some inflammatory cytokines and is also proposed to engage with TLO development. It is likely that TNFα and induced proinflammatory cytokines convert resident stromal fibroblasts into functional LTo cells and initiate lymphoid neogenesis (84) because the ectopic expression of TNFα induces TLO formation in the periphery (63). It was further reported that IL-21, IL-22, and IL-17, produced by Th17, LTi, and γδT cells or neutrophils, are also important players in TLO formation. Increased IL-21 expression has been observed in TLOs in several disease models, such as RA and renal grafts (30, 83, 85). IL-22 promotes TLO development in salivary glands in response to local adenovirus infection (5), and TLO formation in human rheumatoid synovitis is strongly associated with the upregulation of IL-23, IL-21, IL-22, and IL-17F (30).
Chemokines
Chemokines are known to influence leukocyte recruitment and TLO development. CXCL13 is predominantly expressed by fibroblastic stromal cells and regulates B-cell recruitment, differentiation, and maturation. Overexpression of CXCL13 by rat insulin promoter induced TLO formation characterized by T/B-cell zones and HEV (79). In advanced atherosclerosis, activated LTo-like VSMCs highly expressed CXCL13 and CCL21 to induce ATLO neogenesis. CXCL13, CCL21, and CXCL12, were also found in chronic inflammatory diseases, including SS, rheumatoid arthritis, and other disease models (Table 1). As a receptor for CXCL13, CXCR5 is of equal importance for TLO development since TLO has been shown to fail to develop in the absence of CXCR5 (86), indicating that individual chemokines or receptors have a significant impact on TLO development. Accumulating data demonstrated that CXCL13 and LTα1β2 might be the “bio-marker” predicting the formation of TLOs in some diseases, such as RA, SS, and atherosclerosis (87, 88).
CXCL12 is expressed by bone marrow stromal cells, SMCs, and HEVs in lymphoid organs. Transgenic mice with CXCL12 overexpression by RIP showed enriched infiltration of T and B cells, DCs, and plasma cells (89). Increased CXCL12 expression was detected in TLOs in the salivary glands of patients with SS (67). CCL19 and CCL21 are expressed by stromal cells and endothelial cells and interact with CCR7 to regulate T-cell homing during TLO neogenesis. Significant upregulation of CCL19 and CCL21 is observed in ectopic infiltrates of RA and SS (90), whereas CCL21 is more effective than CCL19 in forming TLOs (89). However, it was shown that CXCL12 alone could not promote TLO formation due to its inability to induce LTα1β2 expression (89), indicating that certain chemokines are not sufficient to drive the complete process of TLO formation alone. It might be that different chemokines have a differential capacity to recruit and maintain LTi cells and promote LTα1β2 expression, and they thus showed different abilities to promote TLO development (91).
Taken together, though many cytokines, chemokines, LTs, and receptors have been demonstrated for their roles in TLO development, it is inaccurate to claim that all of them could be used as the biomarkers for TLO formation because most of them perform the TLO-initiating function only in some specific models and local microenvironments. Nevertheless, CXCL13 and LTα1β2 could be candidate molecules that can be considered as potential biomarkers for TLO development.
The Role of LTi Cells in TLO Development
LTi cells, one of two subgroups of type 3 innate lymphoid cells, are hematopoietic cells that were identified in fetal lymphoid tissues. They are critical for the induction of fetal lymphoid tissues, including LNs and Peyer's patches (92). LTi cells can be identified as CD45+CD3−CD4+ cells with a high expression of LTα1β2, RORγt, and IL-7Rα. One of the earliest events in secondary lymphoid organogenesis is the interaction between LTi cells and stromal LTo cells in the LN anlagen through LTβR signaling. Although LTi cells are known to be involved in the development of SLOs, the evidence for the requirement of LTi for TLO induction is still controversial as both LTi-dependent and independent TLO neogenesis have been reported.
Some evidence favors the contribution of LTi cells in the development of TLOs. Meier and colleagues reported that transgenic mice with IL-7 overexpression showed significantly increased amounts of LTi cells in the spleen and organized TLOs in the pancreas and salivary gland, suggesting that IL-7 stimulated LTi cells are essential for TLO development (70). In the CXCL13 transgenic model, the absence of LTi cells results in smaller and less organized cell aggregation, indicating the role of LTi cells in the development of TLOs (87). In human non-small cell lung cancer, tumor NCR+ ILC3 might contribute to tumor-associated TLOs by interacting with both tumor cells and tumor fibroblasts (93). Enhanced lymphocyte infiltrations in rheumatoid arthritis synovial fluid and murine intestine are believed to correlate with increased expression of TNF-related activation-induced cytokine by LTi-like cells (94, 95). Bone marrow derived M1 macrophages has been reported to act as LTi cells and crosstalk with VSMCs, and then initiated TLO formation in a LTβR-independent manner (96).
By contrast, some reports shed negative light on the role of LTi in TLO formation. Deletion of transcription factors, such as inhibitors of DNA-binding 2 (Id2) and ROR-γt, that are known to be critical for LTi-cell differentiation from lymphoid progenitors, lead to complete loss of LTi cells, LNs, and Peyer's patches but did not affect the spleen (97–99). However, when infected with influenza virus, the Id2−/− or Rorc−/− mice developed iBALT in the lung (10). Moreover, mice with overexpression of CCL21 in the thyroid developed ectopic lymphoid tissues, but, when they were crossed with Id2−/− mice lacking LTi cells, the TLO formation was not interrupted (100). Importantly, Schropp et al. revealed that Th17 cells can act as substitutes for LTi cells and contribute to B-cell aggregate formation in the cerebellum in an experimental autoimmune encephalitis mouse model (101). These data indicated that LTi cells might be involved in but might not be essential for the formation of TLO. It is now gradually accepted that, during the chronic inflammatory condition, the TLO-inducing signals can be provided by cell types other than LTi cells (102) because lymphocytes, like B and T cells, are an alternative source of LT when appropriately stimulated (69).
The Role of Stromal Fibroblast Cells in TLO Development
The adult stromal cells are derived from embryonic mesenchymal LTos and are considered to be structural components of an organ. They include fibroblasts, VSMCs, pericytes, epithelial cells, and blood and lymphatic endothelial cells (1). As traditionally considered, stromal cells not only make the architecture of the organ but also regulate the tissue function. Stromal fibroblasts are likely to act as scaffolds for the tissue due to their capacity to synthesize and remodel the extracellular matrix, and these cells in TLOs can be detected using specific marker combinations, such as gp38/CCL21 for FRCs and CD35/CXCL13 for FDCs. FRCs and FDCs in TLOs are predominantly associated with T- and B-cell recruitment, respectively. Therefore, fibroblasts closely interact with other cells to participate in tissue development, differentiation, and repair in the local microenvironment.
A key step in TLO formation is the “switching” of activated stromal cells toward an LTo-like phenotype. An increasing amount of evidence indicates that stromal cells are involved in orchestrating local immune responses and affecting disease progression (1, 63). During TLO neogenesis, tissue-resident stromal cells that gain the function of TLo-like fibroblasts represent a hallmark feature of TLOs due to the expression of lymph organogenic chemokines, such as CXCL13, CCL21, CXCL1, CXCL8, and CCL5 (1, 63). Adipose tissue-derived PDGFRβ+ stromal vascular cells have the capacity to differentiate into FDCs (103), while resident fibroblasts have been shown to massively proliferate and give rise to lymphoid stromal cells during inflammation and ontogeny (78). We previously proved that aortic VSMCs acted as LTo cells and highly expressed CXCL13, CCL21, and LTβR during long-term chronic inflammation in aged Apoe−/− mice (16, 20). In humans, a gp38+ fibroblast showed phenotypical characteristics that were similar to FRC in SS patients, and CXCL13+ stromal cells were detected in synovial tissues of RA patients (38, 87). In addition, FAPα+ fibroblasts promoted inflammation and bone erosions in an arthritis mouse model (104), and Pdpn+ fibroblast were pivotal drivers of TLO formation independent of LT and RORγt (105). Guedj et al. found that mesenteric adipocytes orchestrated the development of functional TLOs in Crohn's disease-affected mesentery (3), suggesting that adipocytes may function as LTo-like cells and promote TLO formation. These data demonstrated that stromal fibroblast cells underwent a complex phenotypical change and acquired LTo-like characteristics for organizing local immune responses.
Emerging evidence indicates that stromal fibroblast cell differentiation is dependent on TNF family members, such as LTα and TNFα. LTα plays a significant role in lymphatic vessel function and in inflammation-associated lymphangiogenesis (106). LTα−/− mice lacking LTα3 and LTα1β2 showed the absence of all LNs and PPs and the disrupted spleen architecture (107). LTβ−/− mice, meanwhile, showed defects in LN development and spleen structure that were less pronounced than that in LTα−/− mice (108). Furtado et al. showed that TNFα signaling pathways were sufficient to induce TLOs in the intestine independently of LTi cells, indicating that interactions between TNFα-expressing myeloid cells and stromal cells were enough to induce TLOs formation (61). IL-17A induced iBALT formation in the lung by directly stimulating the differentiation of CXCL13- and CCL21-expressing stromal cells (51). Furthermore, IL-23 and IL-22 are known to activate fibroblasts from the lung and salivary gland, respectively, to express CXCL13 for TLO formation (5, 80). These data indicated that TNF, LT, and some leukocytes, including myeloid cells and granulocytes that release pro-inflammatory cytokine, are capable of activating resident fibroblasts for their transdifferentiation (5).
TLOs in Diseases
The potential impacts of TLOs on disease progression are increasingly gaining recognition. Autoimmune diseases are abnormal conditions with both B- and T-cell responses against self-tissue antigens. TLOs have been witnessed in various autoimmune diseases, especially in the affected areas, like joints of rheumatoid arthritis (28), meninges in multiple sclerosis (109, 110), salivary glands in SS (111), pancreas in diabetes (112), and the thyroid in Hashimoto's thyroiditis (113) (Table 1). The formation of TLOs in autoimmune diseases may contribute to disease manifestation.
TLOs with activated GCs also expressed the enzyme AID, which mediates B-cell clonal expansion and somatic hypermutation of the V(H) gene within TLOs. Plasma cells then release high-affinity antibodies, such as auto-antibodies targeting ribonucleo proteins Ro and La (Sjogren's syndrome antigen A and Sjogren's syndrome antigen B, respectively) in SS (76). During this process, generated autoreactive B cells in TLO GCs could escape negative selection and apoptosis. For instance, GC B cells from TLOs of SS demonstrate increased anti-apoptotic BCL-2 expression and decreased rate of apoptosis (114). These data demonstrate that TLO GCs provide the microenvironment for antigen-specific antibody production. However, the GCs and accumulated disease-specific autoantibodies could induce adaptive immune responses in local tissues, which perpetuate autoimmune diseases. Therefore, targeting surface antigens on B cells has emerged as a promising therapeutic approach. For example, proteasome inhibitors and antibodies against CD138, B-cell maturation antigen (BCMA), and the Signaling Lymphocyte Activation Molecule family (SLAMF) were used in clinical trials (16, 17, 115). Though limited efficiency in clinical practice is seen, a combination of different therapies could bring about improvement in practical terms. In addition, rituximab (a CD20-depleting antibody that targets B cells before the stage of plasma blast cells) may also be a better alternative for treating rheumatoid arthritis.
TLOs also harbor T follicular helper (Tfh) cells that are critical in T-cell dependent B-cell responses and therefore regulate active GC responses in the lymphoid organ. The interaction of ICOS, CD40, and IL-21 on Tfh cells with ICOSL, CD40L, and IL-21R expressed on GC B cells induces the activation, affinity maturation of B cells, and further differentiation of plasma cells in TLOs (77). It was also reported that Tfh-GC B-cell axis was proatherogenic in an Apoe−/− mouse model (39). Therefore, blocking B-Tfh cell interaction through targeting ICOS-ICOSL, CD40-CD40L, or IL-21-IL-21R could significantly affect downstream TLO B-cell activation.
Besides autoimmune diseases, TLOs also develop in some infectious diseases and various cancers (Table 1), and they are generally believed to perform a protective role by skewing the local immune responses toward anti-infection and anti-tumor outcomes. However, the efficiency of the immune responses may change according to the local concentration of viral and tumor antigens. In a long-lasting inflammation, the synthesis of cytokines, chemokines, and tissue factors is constantly enhanced. Ectopically developed TLOs will mount the local immune responses and deteriorate the disease progression. Therefore, the roles of TLOs in diseases are largely dependent on the local conditions, either protective or harmful.
Clinical Implications
TLO formation may affect the disease process because ectopic GCs in TLOs could potentiate local immune responses. As discussed above, TLOs might be protective in certain diseases, such as infectious diseases, cancers, and atherosclerosis, because they can mount an efficient response against antigenic stimuli in tumors or chronic inflammation. However, in other diseases, including autoimmune diseases and transplantation, TLOs might further aggravate disease progression (62, 66). Therefore, promoting or inhibiting TLO development with tailored immune therapy should be decided based on the disease phenotype.
Given the prominent role of LT in TLO development, targeting the LTα1β2-LTβR signaling pathway has already been used extensively to modulate TLO formation. The decoy receptor–LTβR-Ig fusion protein is used widely to interfere with the signaling pathway in the animal model and preclinical trials (116). It should be noted that LTβR-Ig treatment disturbed spleen microarchitecture, including loss of integrity of marginal zones, FDCs, and cell proliferation (14), and this is suggestive of its side effect on SLOs. The administration of LTβR-Ig prevents insulitis at the early stage, reverses insulitis, and disrupts pancreatic lymphoid aggregates at the late stage of the disease in NOD mice (117). In a mouse model of autoimmune sialoadenitis, LTβR-Ig treatment resulted in decreased immune cell infiltration and a significant improvement in the salivary gland function (118). Notably, vascular smooth muscle cell-specific deletion of LTβR in atherosclerotic mice disrupted adventitial TLO formation and aggravated the atherosclerotic plaque size (20). Thus, the detailed mechanism of disease progression still require further investigation.
Importantly, pateclizumab (a monoclonal antibody against LTα) and baminercept (LTβR-IgG1, an inhibitor of both the LTα1β2 and LIGHT pathways) are currently undergoing Phase I and II trials for the treatment of autoimmune diseases (119). Pateclizumab, a humanized monoclonal antibody against LTα, is already verified to be generally safe and well tolerated in clinical trials (119, 120). Unexpectedly, 6 months of baminercept treatment failed to improve glandular dysfunction in patients with primary SS. Although not achieving expected treatment efficacy, the studies revealed, to some extent, the therapeutic effect of baminercept in LT/LIGHT-dependent pathways, suggesting that blocking LTβR signaling might be of therapeutic benefit at earlier stages (121). Both treatments resulted in decreased serum CXCL13 levels in patients with RA, but baminercept treatment was unable to diminish the serum levels of BAFF, LIGHT, or IP-10 compared to the placebo group (119, 120).
The blockade of a TLO-associated chemokine or its receptor is another therapeutic strategy that has been tested in animal models. Chemokine CXCL13, CCL19, and CCL21 and the receptors CXCR5 and CCR7 play central roles in lymphocyte migration during TLO formation. Targeting these molecules might therefore be pivotal for the therapeutic inhibition of TLOs. It has been reported that the blockade of CXCL13 in mouse models reduces glandular inflammation in SS (122) and decreases the severity of collagen-induced arthritis and GC formation in synovial tissues (123). Despite this, TLO-associated chemokine/receptor-based treatments have not yet entered clinical trials.
Furthermore, other therapeutic strategies targeting TLO-associated molecules or pathways have been launched recently, including Tfh-related molecules, i.e., IL-21 and the receptor IL-21R, co-stimulatory molecules ICOS and its ligand ICOSL, and the IL-17 pathway (66). The pharmacological blockade of these targets has been shown to have a potential function in the experimental disease models, some of which have already entered clinical trials (124).
Concluding Remarks
Chronic inflammation promotes neogenesis of TLOs to control local immunity in a feedback manner. The potential impact of TLOs on disease progression, either protective or damaging, is increasingly attracting peoples' attention. Although considerable progress has been made in the understanding of ectopically developed lymphoid organs, many basic questions remain unsolved. Since TLOs are different from simple leukocyte infiltrates in affected tissues of chronic diseases, a globalized standard for defining and identifying TLOs could be fundamental for all future studies. In the meantime, TLOs can serve as a potential therapeutic target in clinical practices where early diagnosis of TLOs and corresponding appropriate therapies are promising. Nevertheless, specific diagnostic approaches, or biomarkers, to predict TLO development at the early stage of disease are still lacking. Extensive studies on cellular and molecular mechanisms of TLO development are also still required. With regards to therapies, all these aspects should be considered before applying systemic drugs to modulate TLO formation as they could have serious side effects on other lymphoid and non-lymphoid tissue compartments.
Author Contributions
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
Funding
This work has been supported by the grants from the China National Natural Science Foundation (No. 81601747 to SL and Nos. 31770983 and 81974249 to DH), the Natural Science Foundation of Hubei Province (No. 2016CFB684 to RZ), and the German Research Council (MO 3052/1-1 to SM).
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1. Buckley CD, Barone F, Nayar S, Benezech C, Caamano J. Stromal cells in chronic inflammation and tertiary lymphoid organ formation. Annu Rev Immunol. (2015) 33:715–45. doi: 10.1146/annurev-immunol-032713-120252
2. Guedj K, Khallou-Laschet J, Clement M, Morvan M, Delbosc S, Gaston AT, et al. Inflammatory micro-environmental cues of human atherothrombotic arteries confer to vascular smooth muscle cells the capacity to trigger lymphoid neogenesis. PLoS ONE. (2014) 9:e116295. doi: 10.1371/journal.pone.0116295
3. Guedj K, Abitbol Y, Cazals-Hatem D, Morvan M, Maggiori L, Panis Y, et al. Adipocytes orchestrate the formation of tertiary lymphoid organs in the creeping fat of Crohn's disease affected mesentery. J Autoimmun. (2019) 103:102281. doi: 10.1016/j.jaut.2019.05.009
4. Tarkowska A, Zaorska-Rajca J, Wypych M, Adamczyk-Szarewicz W. Relation between mean ventricular phases in chronic cor pulmonale measured by radionuclide ventriculography. Eur J Nucl Med. (1990) 16:693–95. doi: 10.1007/BF00998171
5. Barone F, Nayar S, Campos J, Cloake T, Withers DR, Toellner KM, et al. IL-22 regulates lymphoid chemokine production and assembly of tertiary lymphoid organs. Proc Natl Acad Sci USA. (2015) 112:11024–9. doi: 10.1073/pnas.1503315112
6. Jones GW, Hill DG, Jones SA Understanding immune cells in tertiary lymphoid organ development: it is all starting to come together. Front Immunol. (2016) 7:401. doi: 10.3389/fimmu.2016.00401
7. Pikor NB, Astarita JL, Summers-Deluca L, Galicia G, Qu J, Ward LA, et al. Integration of Th17- and lymphotoxin-derived signals initiates meningeal-resident stromal cell remodeling to propagate neuroinflammation. Immunity. (2015) 43:1160–73. doi: 10.1016/j.immuni.2015.11.010
8. Weninger W, Carlsen H S, Goodarzi M, Moazed F, Crowley MA, Baekkevold ES, et al. Naive T cell recruitment to nonlymphoid tissues: a role for endothelium-expressed CC chemokine ligand 21 in autoimmune disease and lymphoid neogenesis. J Immunol. (2003) 170:4638–48. doi: 10.4049/jimmunol.170.9.4638
9. Collison J. Lupus nephritis: novel role for BAFF in tertiary lymphoid neogenesis. Nat Rev Rheumatol. (2017) 13:260. doi: 10.1038/nrrheum.2017.40
10. Lochner M, Ohnmacht C, Presley L, Bruhns P, Si-Tahar M, Sawa S, et al. Microbiota-induced tertiary lymphoid tissues aggravate inflammatory disease in the absence of RORgamma t and LTi cells. J Exp Med. (2011) 208:125–34. doi: 10.1084/jem.20100052
11. Xu X, Han Y, Wang Q, Cai M, Qian Y, Wang X, et al. Characterisation of tertiary lymphoid organs in explanted rejected donor kidneys. Immunol Invest. (2016) 45:38–51. doi: 10.3109/08820139.2015.1085394
12. Ezzat M, El-Gammasy T, Shaheen K, Shokr E. Elevated production of serum B-cell-attracting chemokine-1. (BCA-1/CXCL13) is correlated with childhood-onset lupus disease activity, severity, and renal involvement. Lupus. (2011) 20:845–54. doi: 10.1177/0961203311398513
13. McNamee EN, Masterson JC, Jedlicka P, Collins CB, Williams IR, Rivera-Nieves J. Ectopic lymphoid tissue alters the chemokine gradient, increases lymphocyte retention and exacerbates murine ileitis. Gut. (2013) 62:53–62. doi: 10.1136/gutjnl-2011-301272
14. Grabner R, Lotzer K, Dopping S, Hildner M, Radke D, Beer M, et al. Lymphotoxin beta receptor signaling promotes tertiary lymphoid organogenesis in the aorta adventitia of aged ApoE−/− mice. J Exp Med. (2009) 206:233–48. doi: 10.1084/jem.20080752
15. Reed H O, Wang L, Sonett J, Chen M, Yang J, Li L, et al. Lymphatic impairment leads to pulmonary tertiary lymphoid organ formation and alveolar damage. J Clin Invest. (2019) 129:2514–26. doi: 10.1172/JCI125044
16. Hu D, Yin C, Luo S, Habenicht AJ R, Mohanta S K, Vascular smooth muscle cells contribute to atherosclerosis immunity. Front Immunol. (2019) 10:1101. doi: 10.3389/fimmu.2019.01101
17. Randall T D, Carragher D M, Rangel-Moreno J. Development of secondary lymphoid organs. Annu Rev Immunol. (2008) 26:627–50. doi: 10.1146/annurev.immunol.26.021607.090257
18. Neyt K, Perros F, GeurtsvanKessel C H, Hammad H, Lambrecht BN Tertiary lymphoid organs in infection and autoimmunity. Trends Immunol. (2012) 33:297–305. doi: 10.1016/j.it.2012.04.006
19. Mohanta SK, Yin C, Peng L, Srikakulapu P, Bontha V, Hu D, et al. Artery tertiary lymphoid organs contribute to innate and adaptive immune responses in advanced mouse atherosclerosis. Circ Res. (2014) 114:1772–87. doi: 10.1161/CIRCRESAHA.114.301137
20. Hu D, Mohanta SK, Yin C, Peng L, Ma Z, Srikakulapu P, et al. Artery tertiary lymphoid organs control aorta immunity and protect against atherosclerosis via vascular smooth muscle cell lymphotoxin β receptors. Immunity. (2015) 42:1100–15. doi: 10.1016/j.immuni.2015.05.015
21. Lee HJ, Kim JY, Park IA, Song IH, Yu JH, Ahn JH, et al. Prognostic significance of tumor-infiltrating lymphocytes and the tertiary lymphoid structures in HER2-positive breast cancer treated with adjuvant trastuzumab. Am J Clin Pathol. (2015) 144:278–88. doi: 10.1309/AJCPIXUYDVZ0RZ3G
22. Lee H J, Park IA, Song IH, Shin SJ, Kim JY, Yu JH, Gong G. Tertiary lymphoid structures: prognostic significance and relationship with tumour-infiltrating lymphocytes in triple-negative breast cancer. J Clin Pathol. (2016) 69:422–430. doi: 10.1136/jclinpath-2015-203089
23. Vayrynen JP, Sajanti SA, Klintrup K, Makela J, Herzig KH, Karttunen TJ, et al. Characteristics and significance of colorectal cancer associated lymphoid reaction. Int J Cancer. (2014) 134:2126–35. doi: 10.1002/ijc.28533
24. Willis SN, Mallozzi SS, Rodig SJ, Cronk KM, McArdel SL, Caron T, et al. The microenvironment of germ cell tumors harbors a prominent antigen-driven humoral response. J Immunol. (2009) 182:3310–17. doi: 10.4049/jimmunol.0803424
25. Gu-Trantien C, Loi S, Garaud S, Equeter C, Libin M, de Wind A, et al. CD4(+) follicular helper T cell infiltration predicts breast cancer survival. J Clin Invest. (2013) 123:2873–92. doi: 10.1172/JCI67428
26. Peters A, Pitcher LA, Sullivan JM, Mitsdoerffer M, Acton SE, Franz B, et al. Th17 cells induce ectopic lymphoid follicles in central nervous system tissue inflammation. Immunity. (2011) 35:986–96. doi: 10.1016/j.immuni.2011.10.015
27. Lee Y, Chin RK, Christiansen P, Sun Y, Tumanov AV, Wang J, et al. Recruitment and activation of naive T cells in the islets by lymphotoxin beta receptor-dependent tertiary lymphoid structure. Immunity. (2006) 25:499–509. doi: 10.1016/j.immuni.2006.06.016
28. Humby F, Bombardieri M, Manzo A, Kelly S, Blades MC, Kirkham B, et al. Ectopic lymphoid structures support ongoing production of class-switched autoantibodies in rheumatoid synovium. PLoS Med. (2009) 6:e1. doi: 10.1371/journal.pmed.0060001
29. Corsiero E, Bombardieri M, Manzo A, Bugatti S, Uguccioni M, Pitzalis C. Role of lymphoid chemokines in the development of functional ectopic lymphoid structures in rheumatic autoimmune diseases. Immunol Lett. (2012) 145:62–7. doi: 10.1016/j.imlet.2012.04.013
30. Canete J D, Celis R, Yeremenko N, Sanmarti R, van Duivenvoorde L, Ramirez J, et al. Ectopic lymphoid neogenesis is strongly associated with activation of the IL-23 pathway in rheumatoid synovitis. Arthritis Res Ther. (2015) 17:173. doi: 10.1186/s13075-015-0688-0
31. Corcione A, Casazza S, Ferretti E, Giunti D, Zappia E, Pistorio A, et al. Recapitulation of B cell differentiation in the central nervous system of patients with multiple sclerosis. Proc Natl Acad Sci USA. (2004) 101:11064–9. doi: 10.1073/pnas.0402455101
32. Lehmann-Horn K, Wang SZ, Sagan SA, Zamvil SS, von Budingen HC. B cell repertoire expansion occurs in meningeal ectopic lymphoid tissue. JCI Insight. (2016) 1:e87234. doi: 10.1172/jci.insight.87234
33. Astorri E, Bombardieri M, Gabba S, Peakman M, Pozzilli P, Pitzalis C. Evolution of ectopic lymphoid neogenesis and in situ autoantibody production in autoimmune nonobese diabetic mice: cellular and molecular characterization of tertiary lymphoid structures in pancreatic islets. J Immunol. (2010) 185:3359–68. doi: 10.4049/jimmunol.1001836
34. Alunno A, Ibba-Manneschi L, Bistoni O, Rosa I, Caterbi S, Gerli R, et al. Telocytes in minor salivary glands of primary Sjogren's syndrome: association with the extent of inflammation and ectopic lymphoid neogenesis. J Cell Mol Med. (2015) 19:1689–96. doi: 10.1111/jcmm.12545
35. Salomonsson S, Larsson P, Tengner P, Mellquist E, Hjelmstrom P, Wahren-Herlenius M. Expression of the B cell-attracting chemokine CXCL13 in the target organ and autoantibody production in ectopic lymphoid tissue in the chronic inflammatory disease Sjogren's syndrome. Scand J Immunol. (2002) 55:336–42. doi: 10.1046/j.1365-3083.2002.01058.x
36. Weiss JM, Robinet M, Aricha R, Cufi P, Villeret B, Lantner F, et al. Novel CXCL13 transgenic mouse: inflammation drives pathogenic effect of CXCL13 in experimental myasthenia gravis. Oncotarget. (2016) 7:7550–62. doi: 10.18632/oncotarget.6885
37. Wong CK, Wong PT, Tam LS, Li EK, Chen DP, Lam CW. Elevated production of B cell chemokine CXCL13 is correlated with systemic lupus erythematosus disease activity. J Clin Immunol. (2010) 30:45–52. doi: 10.1007/s10875-009-9325-5
38. Manzo A, Bugatti S, Caporali R, Prevo R, Jackson DG, Uguccioni M, et al. CCL21 expression pattern of human secondary lymphoid organ stroma is conserved in inflammatory lesions with lymphoid neogenesis. Am J Pathol. (2007) 171:1549–62. doi: 10.2353/ajpath.2007.061275
39. Clement M, Guedj K, Andreata F, Morvan M, Bey L, Khallou-Laschet J, et al. Control of the T follicular helper-germinal center B-cell axis by CD8(+) regulatory T cells limits atherosclerosis and tertiary lymphoid organ development. Circulation. (2015) 131:560–70. doi: 10.1161/CIRCULATIONAHA.114.010988
40. Demoor T, Bracke KR, Maes T, Vandooren B, Elewaut D, Pilette C, et al. Role of lymphotoxin-alpha in cigarette smoke-induced inflammation and lymphoid neogenesis. Eur Respir J. (2009) 34:405–16. doi: 10.1183/09031936.00101408
41. Roos AB, Sanden C, Mori M, Bjermer L, Stampfli MR, Erjefalt JS. IL-17A is elevated in end-stage chronic obstructive pulmonary disease and contributes to cigarette smoke-induced lymphoid neogenesis. Am J Respir Crit Care Med. (2015) 191:1232–41. doi: 10.1164/rccm.201410-1861OC
42. Litsiou E, Semitekolou M, Galani IE, Morianos I, Tsoutsa A, Kara P, et al. CXCL13 production in B cells via Toll-like receptor/lymphotoxin receptor signaling is involved in lymphoid neogenesis in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. (2013) 187:1194–202. doi: 10.1164/rccm.201208-1543OC
43. Lin L, Hu X, Zhang H, Hu H. Tertiary lymphoid organs in cancer immunology: mechanisms and the new strategy for immunotherapy. Front Immunol. (2019) 10:1398. doi: 10.3389/fimmu.2019.01398
44. Germain C, Gnjatic S, Tamzalit F, Knockaert S, Remark R, Goc J, et al. Presence of B cells in tertiary lymphoid structures is associated with a protective immunity in patients with lung cancer. Am J Respir Crit Care Med. (2014) 189:832–44. doi: 10.1164/rccm.201309-1611OC
45. Dieu-Nosjean MC, Antoine M, Danel C, Heudes D, Wislez M, Poulot V, et al. Long-term survival for patients with non-small-cell lung cancer with intratumoral lymphoid structures. J Clin Oncol. (2008) 26:4410–17. doi: 10.1200/JCO.2007.15.0284
46. Muniz LR, Pacer ME, Lira SA, Furtado GC. A critical role for dendritic cells in the formation of lymphatic vessels within tertiary lymphoid structures. J Immunol. (2011) 187:828–34. doi: 10.4049/jimmunol.1004233
47. Meshcheryakova A, Tamandl D, Bajna E, Stift J, Mittlboeck M, Svoboda M, et al. B cells and ectopic follicular structures: novel players in anti-tumor programming with prognostic power for patients with metastatic colorectal cancer. PLoS ONE. (2014) 9:e99008. doi: 10.1371/journal.pone.0099008
48. Cipponi A, Mercier M, Seremet T, Baurain JF, Theate I, van den Oord J, et al. Neogenesis of lymphoid structures and antibody responses occur in human melanoma metastases. Cancer Res. (2012) 72:3997–4007. doi: 10.1158/0008-5472.CAN-12-1377
49. Shields JD, Kourtis IC, Tomei AA, Roberts JM, Swartz MA. Induction of lymphoidlike stroma and immune escape by tumors that express the chemokine CCL21. Science. (2010) 328:749–52. doi: 10.1126/science.1185837
50. Sansonno D, Tucci FA, Troiani L, Lauletta G, Montrone M, Conteduca V, et al. Increased serum levels of the chemokine CXCL13 and up-regulation of its gene expression are distinctive features of HCV-related cryoglobulinemia and correlate with active cutaneous vasculitis. Blood. (2008) 112:1620–7. doi: 10.1182/blood-2008-02-137455
51. Rangel-Moreno J, Carragher DM, de la Luz Garcia-Hernandez M, Hwang JY, Kusser K, Hartson L, et al. The development of inducible bronchus-associated lymphoid tissue depends on IL-17. Nat Immunol. (2011) 12:639–46. doi: 10.1038/ni.2053
52. Moyron-Quiroz JE, Rangel-Moreno J, Hartson L, Kusser K, Tighe MP, Klonowski KD, et al. Persistence and responsiveness of immunologic memory in the absence of secondary lymphoid organs. Immunity. (2006) 25:643–54. doi: 10.1016/j.immuni.2006.08.022
53. Moyron-Quiroz JE, Rangel-Moreno J, Kusser K, Hartson L, Sprague F, Goodrich S, et al. Role of inducible bronchus associated lymphoid tissue. (iBALT) in respiratory immunity. Nat Med. (2004) 10:927–34. doi: 10.1038/nm1091
54. Shomer NH, Fox JG, Juedes AE, Ruddle NH. Helicobacter-induced chronic active lymphoid aggregates have characteristics of tertiary lymphoid tissue. Infect Immun. (2003) 71:3572–7. doi: 10.1128/IAI.71.6.3572-3577.2003
55. Kahnert A, Hopken UE, Stein M, Bandermann S, Lipp M, Kaufmann SH. Mycobacterium tuberculosis triggers formation of lymphoid structure in murine lungs. J Infect Dis. (2007) 195:46–54. doi: 10.1086/508894
56. Alsughayyir J, Pettigrew GJ, Motallebzadeh R. Spoiling for a fight: B lymphocytes as initiator and effector populations within tertiary lymphoid organs in autoimmunity and transplantation. Front Immunol. (2017) 8:1639. doi: 10.3389/fimmu.2017.01639
57. Baddoura FK, Nasr IW, Wrobel B, Li Q, Ruddle NH, Lakkis FG. Lymphoid neogenesis in murine cardiac allografts undergoing chronic rejection. Am J Transplant. (2005) 5:510–6. doi: 10.1111/j.1600-6143.2004.00714.x
58. Motallebzadeh R, Rehakova S, Conlon TM, Win TS, Callaghan CJ, Goddard M, et al. Blocking lymphotoxin signaling abrogates the development of ectopic lymphoid tissue within cardiac allografts and inhibits effector antibody responses. FASEB J. (2012) 26:51–62. doi: 10.1096/fj.11-186973
59. Flavell RA, Kratz A, Ruddle NH. The contribution of insulitis to diabetes development in tumor necrosis factor transgenic mice. Curr Top Microbiol Immunol. (1996) 206:33–50. doi: 10.1007/978-3-642-85208-4_3
60. Martin AP, Coronel EC, Sano G, Chen SC, Vassileva G, Canasto-Chibuque C, et al. A novel model for lymphocytic infiltration of the thyroid gland generated by transgenic expression of the CC chemokine CCL21. J Immunol. (2004) 173:4791–8. doi: 10.4049/jimmunol.173.8.4791
61. Furtado GC, Pacer ME, Bongers G, Benezech C, He Z, Chen L, et al. TNFα-dependent development of lymphoid tissue in the absence of RORγt(+) lymphoid tissue inducer cells. Mucosal Immunol. (2014) 7:602–14. doi: 10.1038/mi.2013.79
62. Aloisi F, Pujol-Borrell R. Lymphoid neogenesis in chronic inflammatory diseases. Nat Rev Immunol. (2006) 6:205–17. doi: 10.1038/nri1786
63. Barone F, Gardner D H, Nayar S, Steinthal N, Buckley CD, Luther SA. Stromal fibroblasts in tertiary lymphoid structures: a novel target in chronic inflammation. Front Immunol. (2016) 7:477. doi: 10.3389/fimmu.2016.00477
64. Yin C, Mohanta SK, Srikakulapu P, Weber C, Habenicht AJ. Artery tertiary lymphoid organs: powerhouses of atherosclerosis immunity. Front Immunol. (2016) 7:387. doi: 10.3389/fimmu.2016.00387
65. Lotzer K, Dopping S, Connert S, Grabner R, Spanbroek R, Lemser B, et al. Mouse aorta smooth muscle cells differentiate into lymphoid tissue organizer-like cells on combined tumor necrosis factor receptor-1/lymphotoxin beta-receptor NF-kappaB signaling. Arterioscler Thromb Vasc Biol. (2010) 30:395–402. doi: 10.1161/ATVBAHA.109.191395
66. Pitzalis C, Jones GW, Bombardieri M, Jones SA. Ectopic lymphoid-like structures in infection, cancer and autoimmunity. Nat Rev Immunol. (2014) 14:447–62. doi: 10.1038/nri3700
67. Barone F, Bombardieri M, Rosado MM, Morgan PR, Challacombe SJ, De Vita S, et al. CXCL13, CCL21, and CXCL12 expression in salivary glands of patients with Sjogren's syndrome and MALT lymphoma: association with reactive and malignant areas of lymphoid organization. J Immunol. (2008) 180:5130–40. doi: 10.4049/jimmunol.180.7.5130
68. Leung GA, Cool T, Valencia CH, Worthington A, Beaudin AE, Forsberg EC. The lymphoid-associated interleukin 7 receptor. (IL7R) regulates tissue-resident macrophage development. Development. (2019) 146:dev176180. doi: 10.1242/dev.176180
69. Luther SA, Ansel KM, Cyster JG. Overlapping roles of CXCL13, interleukin 7 receptor alpha, and CCR7 ligands in lymph node development. J Exp Med. (2003) 197:1191–8. doi: 10.1084/jem.20021294
70. Meier D, Bornmann C, Chappaz S, Schmutz S, Otten LA, Ceredig R, et al. Ectopic lymphoid-organ development occurs through interleukin 7-mediated enhanced survival of lymphoid-tissue-inducer cells. Immunity. (2007) 26:643–54. doi: 10.1016/j.immuni.2007.04.009
71. Benezech C, Luu NT, Walker JA, Kruglov AA, Loo Y, Nakamura K, et al. Inflammation-induced formation of fat-associated lymphoid clusters. Nat Immunol. (2015) 16:819–28. doi: 10.1038/ni.3215
72. GeurtsvanKessel CH, Willart MA, Bergen IM, van Rijt LS, Muskens F, Elewaut D, et al. Dendritic cells are crucial for maintenance of tertiary lymphoid structures in the lung of influenza virus-infected mice. J Exp Med. (2009) 206:2339–49. doi: 10.1084/jem.20090410
73. Laskov R, Lancz G, Ruddle NH, McGrath KM, Specter S, Klein T, et al. Production of tumor necrosis factor. (TNF-alpha) and lymphotoxin. (TNF-beta) by murine pre-B and B cell lymphomas. J Immunol. (1990) 144:3424–30.
74. Lazzeri E, Rotondi M, Mazzinghi B, Lasagni L, Buonamano A, Rosati A, et al. High CXCL10 expression in rejected kidneys and predictive role of pretransplant serum CXCL10 for acute rejection and chronic allograft nephropathy. Transplantation. (2005) 79:1215–20. doi: 10.1097/01.TP.0000160759.85080.2E
75. Steinmetz OM, Panzer U, Kneissler U, Harendza S, Lipp M, Helmchen U, et al. BCA-1/CXCL13 expression is associated with CXCR5-positive B-cell cluster formation in acute renal transplant rejection. Kidney Int. (2005) 67:1616–21. doi: 10.1111/j.1523-1755.2005.00244.x
76. Srikakulapu P, Hu D, Yin C, Mohanta SK, Bontha SV, Peng L, et al. Artery tertiary lymphoid organs control multilayered territorialized atherosclerosis B-cell responses in aged ApoE-/- mice. Arterioscler Thromb Vasc Biol. (2016) 36:1174–85. doi: 10.1161/ATVBAHA.115.306983
77. Corsiero E, Delvecchio FR, Bombardieri M, Pitzalis C. B cells in the formation of tertiary lymphoid organs in autoimmunity, transplantation and tumorigenesis. Curr Opin Immunol. (2019) 57:46–52. doi: 10.1016/j.coi.2019.01.004
78. Peduto L, Dulauroy S, Lochner M, Spath GF, Morales MA, Cumano A, et al. Inflammation recapitulates the ontogeny of lymphoid stromal cells. J Immunol. (2009) 182:5789–99. doi: 10.4049/jimmunol.0803974
79. Drayton DL, Liao S, Mounzer RH, Ruddle NH. Lymphoid organ development: from ontogeny to neogenesis. Nat Immunol. (2006) 7:344–53. doi: 10.1038/ni1330
80. Khader SA, Guglani L, Rangel-Moreno J, Gopal R, Junecko BA, Fountain JJ, et al. IL-23 is required for long-term control of Mycobacterium tuberculosis and B cell follicle formation in the infected lung. J Immunol. (2011) 187:5402–7. doi: 10.4049/jimmunol.1101377
81. Fleige H, Ravens S, Moschovakis GL, Bolter J, Willenzon S, Sutter G, et al. IL-17-induced CXCL12 recruits B cells and induces follicle formation in BALT in the absence of differentiated FDCs. J Exp Med. (2014) 211:643–51. doi: 10.1084/jem.20131737
82. Lee JJ, McGarry MP, Farmer SC, Denzler KL, Larson KA, Carrigan PE, et al. Interleukin-5 expression in the lung epithelium of transgenic mice leads to pulmonary changes pathognomonic of asthma. J Exp Med. (1997) 185:2143–56. doi: 10.1084/jem.185.12.2143
83. Jones GW, Bombardieri M, Greenhill CJ, McLeod L, Nerviani A, Rocher-Ros V, et al. Interleukin-27 inhibits ectopic lymphoid-like structure development in early inflammatory arthritis. J Exp Med. (2015) 212:1793–802. doi: 10.1084/jem.20132307
84. Brembilla NC, Dufour AM, Alvarez M, Hugues S, Montanari E, Truchetet ME, et al. IL-22 capacitates dermal fibroblast responses to TNF in scleroderma. Ann Rheum Dis. (2016) 75:1697–705. doi: 10.1136/annrheumdis-2015-207477
85. Deteix C, Attuil-Audenis V, Duthey A, Patey N, McGregor B, Dubois V, et al. Intragraft Th17 infiltrate promotes lymphoid neogenesis and hastens clinical chronic rejection. J Immunol. (2010) 184:5344–51. doi: 10.4049/jimmunol.0902999
86. Winter S, Loddenkemper C, Aebischer A, Rabel K, Hoffmann K, Meyer TF, et al. The chemokine receptor CXCR5 is pivotal for ectopic mucosa-associated lymphoid tissue neogenesis in chronic Helicobacter pylori-induced inflammation. J Mol Med. (2010) 88:1169–80. doi: 10.1007/s00109-010-0658-6
87. Link A, Hardie DL, Favre S, Britschgi MR, Adams DH, Sixt M, et al. Association of T-zone reticular networks and conduits with ectopic lymphoid tissues in mice and humans. Am J Pathol. (2011) 178:1662–75. doi: 10.1016/j.ajpath.2010.12.039
88. Weyand CM, Goronzy JJ. Ectopic germinal center formation in rheumatoid synovitis. Ann N Y Acad Sci. (2003) 987:140–9. doi: 10.1111/j.1749-6632.2003.tb06042.x
89. Luther SA, Bidgol A, Hargreaves DC, Schmidt A, Xu Y, Paniyadi J, et al. Differing activities of homeostatic chemokines CCL19, CCL21, and CXCL12 in lymphocyte and dendritic cell recruitment and lymphoid neogenesis. J Immunol. (2002) 169:424–33. doi: 10.4049/jimmunol.169.1.424
90. Manzo A, Paoletti S, Carulli M, Blades MC, Barone F, Yanni G, et al. Systematic microanatomical analysis of CXCL13 and CCL21 in situ production and progressive lymphoid organization in rheumatoid synovitis. Eur J Immunol. (2005) 35:1347–59. doi: 10.1002/eji.200425830
91. Mueller CG, Nayar S, Campos J, Barone F. Molecular and cellular requirements for the assembly of tertiary lymphoid structures. Adv Exp Med Biol. (2018) 1060:55–72. doi: 10.1007/978-3-319-78127-3_4
92. Withers DR. Lymphoid tissue inducer cells. Curr Biol. (2011) 21:R381–2. doi: 10.1016/j.cub.2011.03.022
93. Carrega P, Loiacono F, Di Carlo E, Scaramuccia A, Mora M, Conte R, et al. NCR(+)ILC3 concentrate in human lung cancer and associate with intratumoral lymphoid structures. Nat Commun. (2015) 6:8280. doi: 10.1038/ncomms9280
94. Koo J, Kim S, Jung WJ, Lee YE, Song GG, Kim KS, et al. Increased lymphocyte infiltration in rheumatoid arthritis is correlated with an increase in LTi-like cells in synovial fluid. Immune Netw. (2013) 13:240–8. doi: 10.4110/in.2013.13.6.240
95. Marchesi F, Martin AP, Thirunarayanan N, Devany E, Mayer L, Grisotto MG, et al. CXCL13 expression in the gut promotes accumulation of IL-22-producing lymphoid tissue-inducer cells, and formation of isolated lymphoid follicles. Mucosal Immunol. (2009) 2:486–94. doi: 10.1038/mi.2009.113
96. Guedj K, Khallou-Laschet J, Clement M, Morvan M, Gaston AT, Fornasa G, et al. M1 macrophages act as LTβR-independent lymphoid tissue inducer cells during atherosclerosis-related lymphoid neogenesis. Cardiovasc Res. (2014) 101:434–43. doi: 10.1093/cvr/cvt263
97. Yokota Y, Mansouri A, Mori S, Sugawara S, Adachi S, Nishikawa S, et al. Development of peripheral lymphoid organs and natural killer cells depends on the helix-loop-helix inhibitor Id2. Nature. (1999) 397:702–6. doi: 10.1038/17812
98. Kurebayashi S, Ueda E, Sakaue M, Patel DD, Medvedev A, Zhang F, et al. Retinoid-related orphan receptor gamma. (RORgamma) is essential for lymphoid organogenesis and controls apoptosis during thymopoiesis. Proc Natl Acad Sci USA. (2000) 97:10132–7. doi: 10.1073/pnas.97.18.10132
99. Eberl G, Marmon S, Sunshine MJ, Rennert PD, Choi Y, Littman DR. An essential function for the nuclear receptor RORgamma(t) in the generation of fetal lymphoid tissue inducer cells. Nat Immunol. (2004) 5:64–73. doi: 10.1038/ni1022
100. Marinkovic T, Garin A, Yokota Y, Fu YX, Ruddle NH, Furtado GC, et al. Interaction of mature CD3+CD4+ T cells with dendritic cells triggers the development of tertiary lymphoid structures in the thyroid. J Clin Invest. (2006) 116:2622–32. doi: 10.1172/JCI28993.
101. Schropp V, Rohde J, Rovituso DM, Jabari S, Bharti R, Kuerten S. Contribution of LTi and TH17 cells to B cell aggregate formation in the central nervous system in a mouse model of multiple sclerosis. J Neuroinflam. (2019) 16:111. doi: 10.1186/s12974-019-1500-x
102. Nagatake T, Fukuyama S, Kim DY, Goda K, Igarashi O, Sato S, et al. Id2-, RORγt-, and LTβR-independent initiation of lymphoid organogenesis in ocular immunity. J Exp Med. (2009) 206:2351–64. doi: 10.1084/jem.20091436
103. Krautler NJ, Kana V, Kranich J, Tian Y, Perera D, Lemm D, et al. Follicular dendritic cells emerge from ubiquitous perivascular precursors. Cell. (2012) 150:194–206. doi: 10.1016/j.cell.2012.05.032
104. Croft AP, Campos J, Jansen K, Turner JD, Marshall J, Attar M, et al. Distinct fibroblast subsets drive inflammation and damage in arthritis. Nature. (2019) 570:246–51. doi: 10.1038/s41586-019-1263-7
105. Nayar S, Campos J, Smith CG, Iannizzotto V, Gardner DH, Mourcin F, et al. Immunofibroblasts are pivotal drivers of tertiary lymphoid structure formation and local pathology. Proc Natl Acad Sci USA. (2019) 116:13490–7. doi: 10.1073/pnas.1905301116
106. Mounzer RH, Svendsen OS, Baluk P, Bergman CM, Padera TP, Wiig H, et al. Lymphotoxin-alpha contributes to lymphangiogenesis. Blood. (2010) 116:2173–82. doi: 10.1182/blood-2009-12-256065
107. De Togni P, Goellner J, Ruddle NH, Streeter PR, Fick A, Mariathasan S, et al. Abnormal development of peripheral lymphoid organs in mice deficient in lymphotoxin. Science. (1994) 264:703–7. doi: 10.1126/science.8171322
108. Alimzhanov MB, Kuprash DV, Kosco-Vilbois MH, Luz A, Turetskaya RL, Tarakhovsky A, et al. Abnormal development of secondary lymphoid tissues in lymphotoxin beta-deficient mice. Proc Natl Acad Sci USA. (1997) 94:9302–7. doi: 10.1073/pnas.94.17.9302
109. Magliozzi R, Howell O, Vora A, Serafini B, Nicholas R, Puopolo M, et al. Meningeal B-cell follicles in secondary progressive multiple sclerosis associate with early onset of disease and severe cortical pathology. Brain. (2007) 130:1089–104. doi: 10.1093/brain/awm038
110. Londono AC, Mora CA. Role of CXCL13 in the formation of the meningeal tertiary lymphoid organ in multiple sclerosis. F1000Res. (2018) 7:514. doi: 10.12688/f1000research.14556.1
111. Salomonsson S, Jonsson MV, Skarstein K, Brokstad KA, Hjelmstrom P, Wahren-Herlenius M, et al. Cellular basis of ectopic germinal center formation and autoantibody production in the target organ of patients with Sjogren's syndrome. Arthritis Rheum. (2003) 48:3187–201. doi: 10.1002/art.11311
112. Ludewig B, Odermatt B, Landmann S, Hengartner H, Zinkernagel RM. Dendritic cells induce autoimmune diabetes and maintain disease via de novo formation of local lymphoid tissue. J Exp Med. (1998) 188:1493–501. doi: 10.1084/jem.188.8.1493
113. Lira SA, Martin AP, Marinkovic T, Furtado GC. Mechanisms regulating lymphocytic infiltration of the thyroid in murine models of thyroiditis. Crit Rev Immunol. (2005) 25:251–62. doi: 10.1615/CritRevImmunol.v25.i4.10
114. Szodoray P, Jellestad S, Teague MO, Jonsson R. Attenuated apoptosis of B cell activating factor-expressing cells in primary Sjogren's syndrome. Lab Invest. (2003) 83:357–65. doi: 10.1097/01.LAB.0000059930.92336.E2
115. Hiepe F, Radbruch A. Plasma cells as an innovative target in autoimmune disease with renal manifestations. Nat Revi Nephrol. (2016) 12:232–40. doi: 10.1038/nrneph.2016.20
116. Gommerman JL, Browning JL. Lymphotoxin/light, lymphoid microenvironments and autoimmune disease. Nat Rev Immunol. (2003) 3:642–55. doi: 10.1038/nri1151
117. Wu Q, Salomon B, Chen M, Wang Y, Hoffman LM, Bluestone JA, et al. Reversal of spontaneous autoimmune insulitis in nonobese diabetic mice by soluble lymphotoxin receptor. J Exp Med. (2001) 193:1327–32. doi: 10.1084/jem.193.11.1327
118. Gatumu MK, Skarstein K, Papandile A, Browning JL, Fava RA, et al. Blockade of lymphotoxin-beta receptor signaling reduces aspects of Sjogren's syndrome in salivary glands of non-obese diabetic mice. Arthritis Res Ther. (2009) 11:R24. doi: 10.1186/ar2617
119. Emu B, Luca D, Offutt C, Grogan JL, Rojkovich B, Williams MB, et al. Safety, pharmacokinetics, and biologic activity of pateclizumab, a novel monoclonal antibody targeting lymphotoxin α: results of a phase I randomized, placebo-controlled trial. Arthritis Res Ther. (2012) 14:R6. doi: 10.1186/ar3554
120. Kennedy WP, Simon JA, Offutt C, Horn P, Herman A, Townsend MJ, et al. Efficacy and safety of pateclizumab. (anti-lymphotoxin-α) compared to adalimumab in rheumatoid arthritis: a head-to-head phase 2 randomized controlled study. (The ALTARA Study). Arthritis Res Ther. (2014) 16:467. doi: 10.1186/s13075-014-0467-3
121. St.Clair EW, Baer AN, Wei C, Noaiseh G, Parke A, Coca A, et al. Autoimmunity centers of, clinical efficacy and safety of baminercept, a lymphotoxin β receptor fusion protein, in primary Sjögren's syndrome: results from a phase ii randomized, double-blind, placebo-controlled trial. Arthr Rheumatol. (2018) 70:1470–80. doi: 10.1002/art.40513
122. Kramer JM, Klimatcheva E, Rothstein TL. CXCL13 is elevated in Sjogren's syndrome in mice and humans and is implicated in disease pathogenesis. J Leukoc Biol. (2013) 94:1079–89. doi: 10.1189/jlb.0113036
123. Zheng B, Ozen Z, Zhang X, De Silva S, Marinova E, Guo L, et al. CXCL13 neutralization reduces the severity of collagen-induced arthritis. Arthr Rheum. (2005) 52:620–6. doi: 10.1002/art.20768
Keywords: immunity, inflammation, atherosclerosis, tertiary lymphoid organs, adventitia
Citation: Luo S, Zhu R, Yu T, Fan H, Hu Y, Mohanta SK and Hu D (2019) Chronic Inflammation: A Common Promoter in Tertiary Lymphoid Organ Neogenesis. Front. Immunol. 10:2938. doi: 10.3389/fimmu.2019.02938
Received: 15 June 2019; Accepted: 29 November 2019;
Published: 18 December 2019.
Edited by:
Daniela Bosisio, University of Brescia, ItalyReviewed by:
Elena Pipi, Birmingham College, United KingdomYolande Richard, Institut National de la Santé et de la Recherche Médicale (INSERM), France
Copyright © 2019 Luo, Zhu, Yu, Fan, Hu, Mohanta and Hu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Desheng Hu, desheng.hu@hust.edu.cn
 Shanshan Luo
Shanshan Luo Rui Zhu
Rui Zhu Ting Yu
Ting Yu Heng Fan2
Heng Fan2 Sarajo Kumar Mohanta
Sarajo Kumar Mohanta Desheng Hu
Desheng Hu