Gelatin-based hydrogels as versatile tools for tissue engineering: insights from multimodal imaging in vivo and ex vivo
Christoph
Tondera1, 2*,
Sandra
Ullm1, 2*,
Anne
Krüger-Genge3*,
Friedrich
Jung3, 4*,
Tim
P.
Gebauer3, 4*,
Axel
T.
Neffe3, 4*,
Andreas
Lendlein3, 4*,
Jörg
Steinbach1, 2* and
Jens
Pietzsch1, 2*
-
1
Helmholtz-Zentrum Dresden-Rossendorf, Department Radiopharmaceutical and Chemical Biology, Germany
-
2
Technische Universität Dresden, Department of Chemistry and Food Chemistry, Germany
-
3
Helmholtz-Zentrum Geesthacht, Institute of Biomaterial Science and Berlin Brandenburg Centre for Regenerative Therapies, Germany
-
4
Helmholtz Virtual Institute, Multifunctional Biomaterials for Medicin, Germany
Hydrogels based on gelatin have evolved as promising multifunctional biomaterials. Gelatin chains offer cell adhesion sites and suitable degradation characteristics accompanied by low or no toxicity and immunogenicity, respectively. The molar ratio of gelatin and ethyl lysine diisocyanate (LDI) in the starting material mixture determines the elastic properties of the resulting material consisting only of amino acids which are connected by amide and urea junction units.
In order to investigate the potential of these hydrogels in a preclinical model, two gelatin-based hydrogels with different ratios of gelatin and diisocyanate (3-fold molar excess (G10_LNCO3) and 8-fold molar excess (G10_LNCO8) of isocyanate groups), which were formerly characterized in vitro [1], were subcutaneously implanted in immunocompetent SKH-1 nude mice. The observation period covered 84 days post implantation (3 groups (single-implanted hydrogels vs. sham-operated); 6-8 time points; 3-7 animals each). In vivo, hydrogel degradation was followed by magnetic resonance imaging at 7 Tesla with a T2-weighted sequence and applying a voxel-based evaluation method for volume determination. Furthermore, matrix metalloproteinase (MMP) activity was measured using optical imaging after administration of a specifically activatable fluorescent probe and ex vivo using a fluorescent substrate. Moreover, serum analysis of cytokines and MMP levels was performed. Ex vivo, histological staining of different cell types (vascular cells, pan/M2 macrophages, fibroblasts) served for analysis of hydrogel-tissue interaction on a cellular scale.
G10_LNCO8 degraded slower than G10_LNCO3 from day 14 onwards [Figure 1], which can be rationalized by a higher number of netpoints in the former networks.
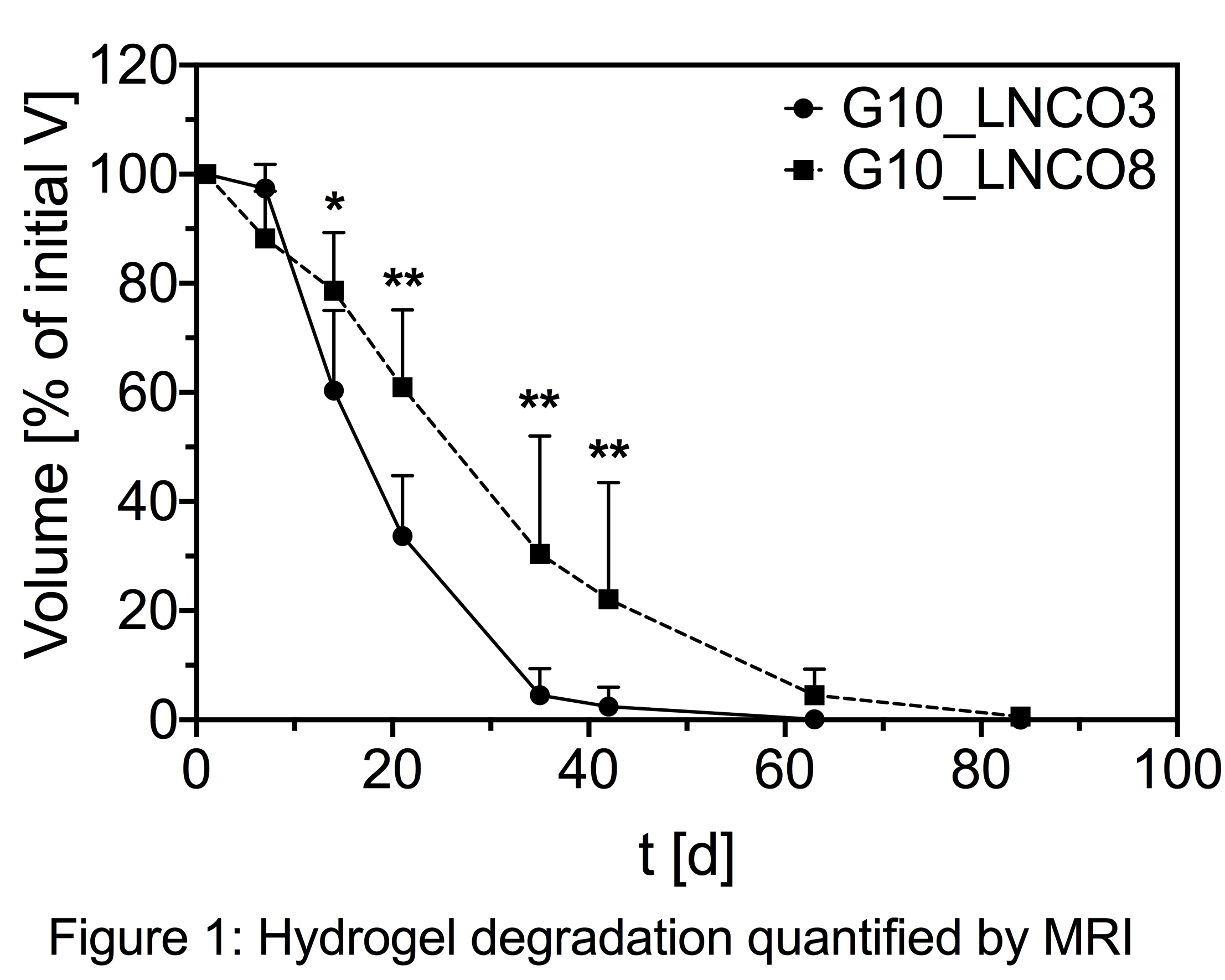
G10_LNCO3 hydrogels were almost completely degraded (V < 5% Vday 1) after 5 weeks while G10_LNCO8 still remained in the body for at least 8 weeks. Both hydrogels resulted in similar local intensity maxima of MMP activity. However, intensity peaked at day 7 for G10_LNCO3 compared to day 14 for G10_LNCO8 [Figure 2].
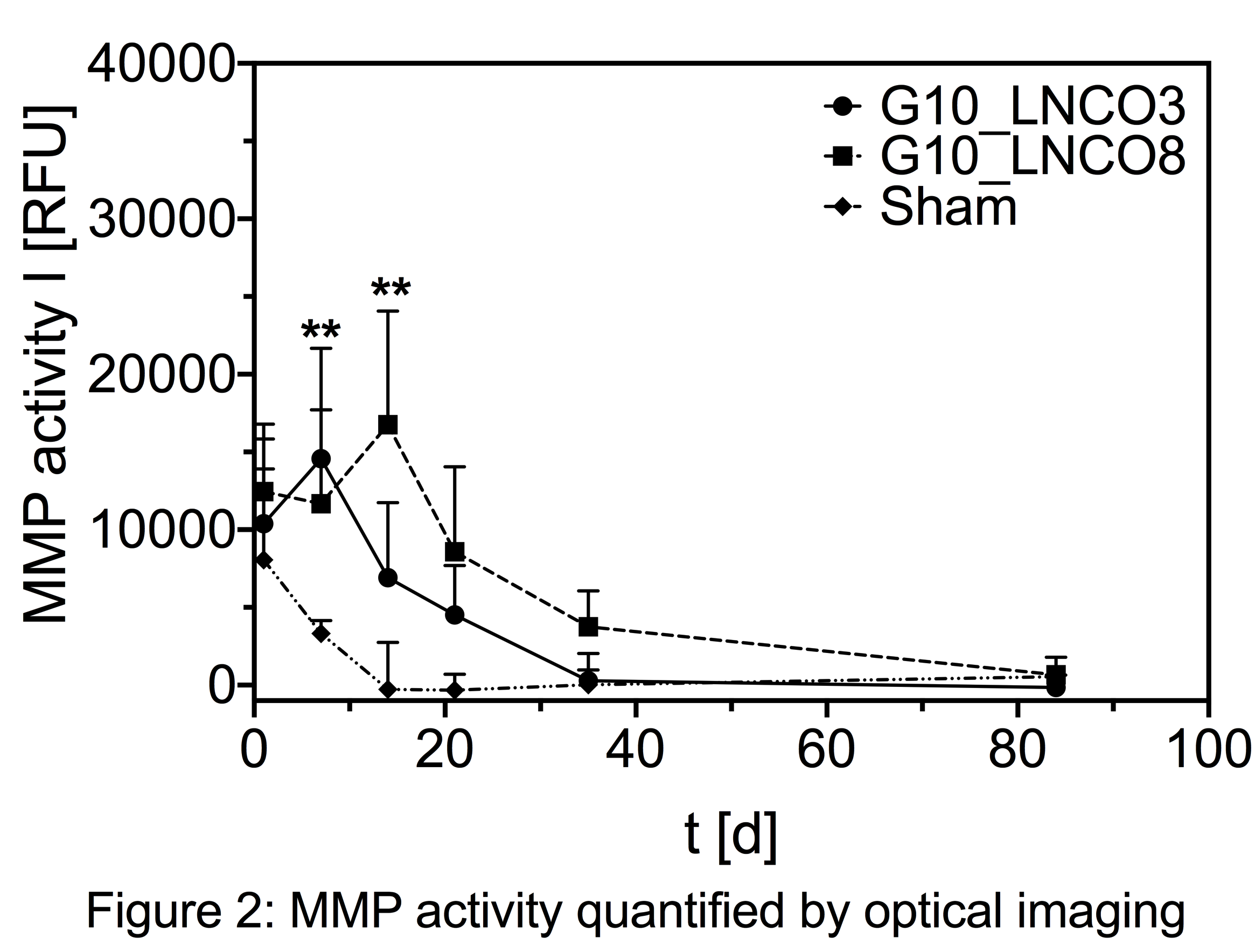
Histological examinations revealed differences in accumulation of macrophages with a time course similar to local MMP activity. M2 macrophages showed no significant increase compared to control animals (sham-operated). Proliferation of blood vessels and fibroblasts was activated by both hydrogels from day 14 till day 21. Substantial sprouting of new blood vessels directly reaching the hydrogel-tissue contact site prevented formation of an impermeable capsule. Implantation of hydrogels caused no systemic effects on cytokine and MMP levels as well as MMP activity.
This multimodal imaging study revealed the diisocyanate content of the investigated gelatin-based hydrogels to correlate well with degradation time. This allows for targeted modification of hydrogels according to the respective tissue to be replaced. As one reason for the different degradation time the time course of macrophage accumulation was identified. Histological investigations of other cell types and serum analytes revealed local mechanisms of tissue remodeling due to degradation, but no adverse local or systemic effects. Finally, this preclinical investigation in immunocompetent mice demonstrated a visible restoration of the original healthy condition.
References:
[1] Ullm, S. et al. Biomaterials 35, 9755–9766 (2014)
Keywords:
Hydrogel,
Molecular Imaging,
MRI,
Biodegradable material
Conference:
10th World Biomaterials Congress, Montréal, Canada, 17 May - 22 May, 2016.
Presentation Type:
Poster
Topic:
Biomaterials evaluation in animal models
Citation:
Tondera
C,
Ullm
S,
Krüger-Genge
A,
Jung
F,
Gebauer
TP,
Neffe
AT,
Lendlein
A,
Steinbach
J and
Pietzsch
J
(2016). Gelatin-based hydrogels as versatile tools for tissue engineering: insights from multimodal imaging in vivo and ex vivo.
Front. Bioeng. Biotechnol.
Conference Abstract:
10th World Biomaterials Congress.
doi: 10.3389/conf.FBIOE.2016.01.01428
Copyright:
The abstracts in this collection have not been subject to any Frontiers peer review or checks, and are not endorsed by Frontiers.
They are made available through the Frontiers publishing platform as a service to conference organizers and presenters.
The copyright in the individual abstracts is owned by the author of each abstract or his/her employer unless otherwise stated.
Each abstract, as well as the collection of abstracts, are published under a Creative Commons CC-BY 4.0 (attribution) licence (https://creativecommons.org/licenses/by/4.0/) and may thus be reproduced, translated, adapted and be the subject of derivative works provided the authors and Frontiers are attributed.
For Frontiers’ terms and conditions please see https://www.frontiersin.org/legal/terms-and-conditions.
Received:
27 Mar 2016;
Published Online:
30 Mar 2016.
*
Correspondence:
Dr. Christoph Tondera, Helmholtz-Zentrum Dresden-Rossendorf, Department Radiopharmaceutical and Chemical Biology, Dresden, Germany, c.tondera@hzdr.de
Dr. Sandra Ullm, Helmholtz-Zentrum Dresden-Rossendorf, Department Radiopharmaceutical and Chemical Biology, Dresden, Germany, s.ullm@hzdr.de
Dr. Anne Krüger-Genge, Helmholtz-Zentrum Geesthacht, Institute of Biomaterial Science and Berlin Brandenburg Centre for Regenerative Therapies, Teltow, Germany, Anne.Krueger@hzg.de
Dr. Friedrich Jung, Helmholtz-Zentrum Geesthacht, Institute of Biomaterial Science and Berlin Brandenburg Centre for Regenerative Therapies, Teltow, Germany, Friedrich.Jung@hzg.de
Dr. Tim P Gebauer, Helmholtz-Zentrum Geesthacht, Institute of Biomaterial Science and Berlin Brandenburg Centre for Regenerative Therapies, Teltow, Germany, Tim.Gebauer@hzg.de
Dr. Axel T Neffe, Helmholtz-Zentrum Geesthacht, Institute of Biomaterial Science and Berlin Brandenburg Centre for Regenerative Therapies, Teltow, Germany, Axel.Neffe@hzg.de
Dr. Andreas Lendlein, Helmholtz-Zentrum Geesthacht, Institute of Biomaterial Science and Berlin Brandenburg Centre for Regenerative Therapies, Teltow, Germany, andreas.lendlein@hzg.de
Dr. Jörg Steinbach, Helmholtz-Zentrum Dresden-Rossendorf, Department Radiopharmaceutical and Chemical Biology, Dresden, Germany, j.steinbach@hzdr.de
Dr. Jens Pietzsch, Helmholtz-Zentrum Dresden-Rossendorf, Department Radiopharmaceutical and Chemical Biology, Dresden, Germany, j.pietzsch@hzdr.de