Tissue engineering and regenerative medicine aim to develop materials that mimic human tissues with the purpose of replacing them when they are compromised, as for degenerative diseases. Important to this aim is the understanding of processes occurring at the cell-biomaterials interface. Cells can sense and respond to chemical and physical stimuli coming from their surroundings (the extracellular matrix -ECM) ultimately deciding whether to adhere or not to them [1]. Cell adhesion is a complex process which involves recruiting of molecules to focal adhesion clusters and coupling of shear stress at the cell membrane to the actin cytoskeleton, and which activates a cascade of signalling pathways that control cell behaviour and, in the case of stem cells, cell fate [2]. Scientists have modified the surface of materials at the nanoscale via either chemical [3] or topographical [4]-[6] patterning techniques to assess how cells sense these features. Here we developed a novel technique (fibre mask lithography) to produce fibre-shaped quasi 2D nanopatterns with varying sizes and controlled chemistry on large scales. This constrained geometry will help assessing the early stages of the cell adhesion process and cytoskeleton dynamics and giving insight in cell response to extracellular stimuli.
The nanopatterning technique we developed is based on electrospinning of polymeric fibres (which constitute the "mask") and reproduction of this geometry via surface-initiated polymerisation [7]. After the mask removal, the fibre shaped pattern left is selectively functionalised with a cell adhesive protein (fibronectin in this case) while the background presents a non-fouling thin layer of polymer brushes [8]. Cells were then seeded on the pattern and their phenotype studied.
Fibre diameters ranging from 200 nm to 1000 nm were obtained in order to study the adhesion process at different stages, from nascent adhesions (composed of few molecules) to more organised structures composed of hundreds of molecules (focal adhesions), and reorganisation of the cell cytoskeleton. Cells were effectively adhering on the patterned area only after functionalization with fibronectin, proving the effectiveness of the non-fouling background generated (polymer brushes). Cell spreading was found to depend strongly on fibre size, with cells struggling to adhere and spread on the smallest topographies while forming mature adhesions and stress fibres on the wider topographies. It is clear that recruiting of molecules to the adhesion site is essential for cell cytoskeleton assembly and cell shape. When adhesion complexes are not allowed to mature, due to geometrical constraints, stress fibre formation is prevented, resulting in abnormal morphologies and restricted cell spreading.
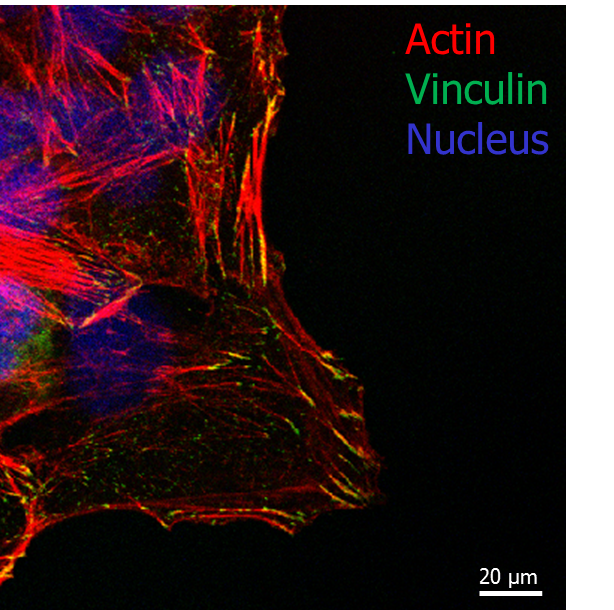
Fig.1 Confocal microscopy image of cell adhering on 500nm fibres diameter pattern: actin (red), vinculin (green) and dapi (blue).
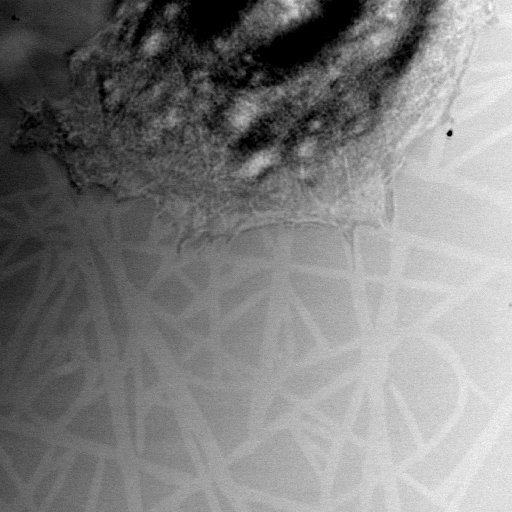
Fig.2 Confocal microscopy image (reflection mode) of cell on pattern.
The development of a novel and scalable nanopattern with a controlled topography and chemistry allowed us to assess how cells behave depending on the constrained geometry. This work showed an effective dependence of cell morphologies on patter size. Future directions of this project will investigate further the fundamental processes underlying cell adhesion to structured surfaces with possible application for tissue engineering and regenerative medicine.
References:
[1] Alexander D. Bershadsky et al., Assembly and mechanosensory function of focal adhesions: experiments and models. European Journal of Cell Biology. 2006 85 165–173
[2] B. Geiger et al., Environmental sensing through focal adhesions. Nature Reviews, 2009, 10, 21-33
[3] Janosch A. Deeg et al., Impact of Local versus Global Ligand Density on Cellular Adhesion. Nano Lett, 2011, 11, 1469-76
[4] Gregory T. Christopherson et al., The influence of fiber diameter of electrospun substrates on neural stem cell differentiation and proliferation. Biomaterials, 2009, 30, 556-64
[5] Matthew J. Dalby et al., The control of human mesenchymal cell differentiation using nanoscale symmetry and disorder. Nat Mater, 2007, 6, 997-1003
[6] Julien E. Gautrot et al., The Nanoscale Geometrical Maturation of Focal Adhesions Controls Stem Cell Differentiation and Mechanotransduction. Nano Lett., 2014, 14, 3945–3952
[7] Mahentha Krishnamoorthy et al., Surface-Initiated Polymer Brushes in the Biomedical Field: Applications in Membrane Science, Biosensing, Cell Culture, Regenerative Medicine and Antibacterial Coatings. Chem. Rev. 2014, 114, 10976−11026
[8] Julien E. Gautrot et al., Exploiting the superior protein resistance of polymer brushes to control single cell adhesion and polarisation at the micron scale. Biomaterials, 2010,31, 5030-5041