Introduction: Functionalized nanoparticles (NPs) can recognize/bind tumor cells and can also be internalized mainly through a receptor-mediated endocytic mechanism. In this frame, hyaluronic acid (HA), a naturally-occurring anionic polysaccharide, has attracted significant research attention for tumor-targeted delivery since it can specifically bind CD44 and RHAMM receptors, which are overexpressed in many forms of cancer[1],[2]. In this context, we propose a novel strategy to directed the self-assembly of HA moieties on NPs based on poly(lactic-co-glycolic acid) (PLGA) and loaded with Irinotecan (IRIN), a sparingly water soluble antitumor drug. The arrangement of HA on NP external surface, with PLGA as the main component of NP bulk, has been directed by creating a lipophilicity gradient between the oil and the water phases of the emulsion used to produce the NPs. The interaction between hydrophilic HA and lipophilic PLGA has been obtained using amphiphilic poloxamers as a bridge between PLGA and HA. The obtained NPs were characterized for their size, morphology and zeta potential. NP uptake and cytotoxicity were assessed on cell line overexpressing CD44 and the effect of HA molecular weight on cell internalization was assessed.
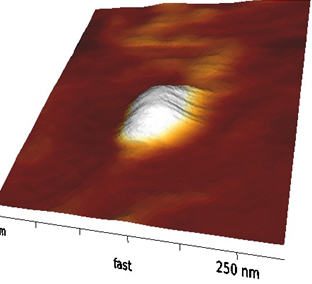
Materials and Methods: NPs were produced by a single emulsion/solvent evaporation technique. Briefly, solutions of PLGA and poloxamers (F127 and F68 1:1 w/w) in acetone, at different PLGA:HA weight ratios, were prepared. The organic phase was emulsified by sonication with an aqueous phase containing different amounts of HA (150 and 850 KDa of molecular weight Mw) and poloxamers. NPs were observed by TEM and AFM, while their size and zeta potential were obtained by Photon Correlation. In vitro drug release profile were studied in phosphate buffer solutions and quantifying the drug by spectrophotometric assay. Finally, differential scanning calorimetry and zeta potential analyses were employed to investigate polymer assembly in NPs. Hs578T and HepG2 cell line and L929 (control) were used. NP cellular uptake was followed by means of a fluorescence microscope while Alamar Blue assay was used to assess NP cytotoxicity.
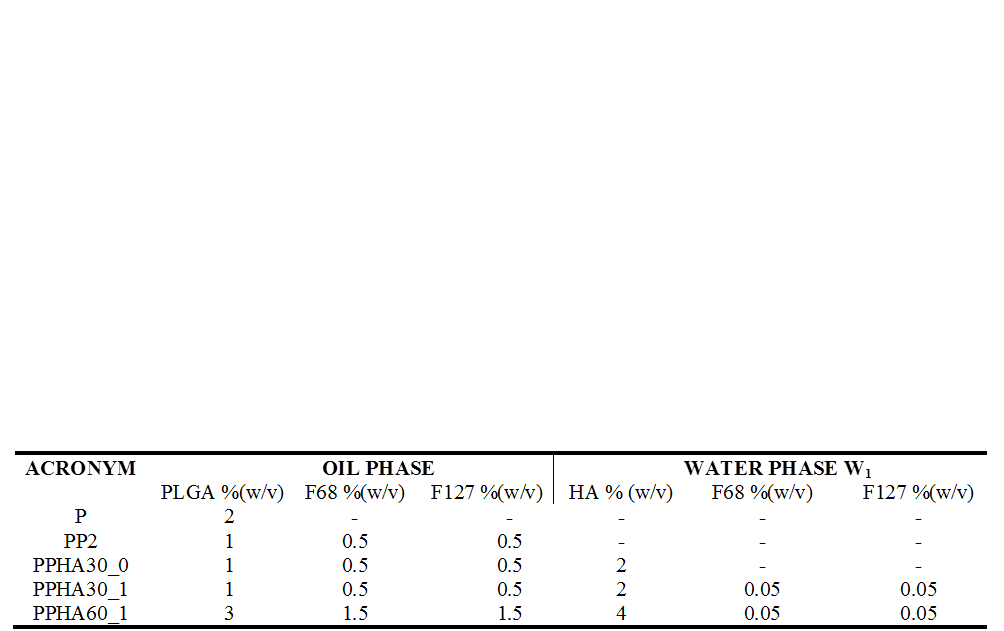
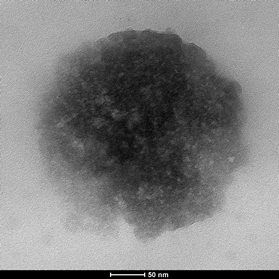
Results and Discussion: NP formulations are defined in Table 1. NPs were spherical and their average size ranged from 100 to 306 nm (Fig.1). Thermal analyses revealed that in the produced NP formulations PLGA, poloxamer and HA act as independent entities and not as a polymeric blend. AFM imaging showed the presence of HA on NP surface (Fig. 2). Together with the results of z potential analyses, this suggests a polymer self-organization driven by a gradient of lipophilicity between the oil and water phases of the emulsion used to prepare the NPs. The HA cover enhances NP size stability over time due to an enhanced electrostatic repulsion of HA-coated NPs and also to a higher degree of hydration and/or to a steric stability caused by the presence of flexible HA chains on NP surface. The amphiphilic nature of these NPs, which possess an inner hydrophobic core and an outer hydrophilic shell, allowed to obtain a IRIN encapsulation efficiency higher than 60%. The in vitro release profiles showed a sustained IRIN release up to 7days. Uptake and cytotoxicity analyses revealed the tropism of HA-coated NPs toward cells overexpressing CD44 receptor which is conditional on the Mw of the HA used to cover NP surface.
Conclusions: Taken all together, the results here collected designate PPHA NPs as promising candidates for targeted drug delivery to liver tumors.
References:
[1] Platt VM, Szoka FC Jr. Anticancer therapeutics: targeting macromolecules and nanocarriers to hyaluronan or CD44, a hyaluronan receptor. Mol Pharm. 2008;5(4):474-86
[2] Lee MY, Yang JA, Jung HS, Beack S, Choi JE, Hur W, Koo H, Kim K, Yoon SK, Hahn SK. Hyaluronic acid-gold nanoparticle/interferon α complex for targeted treatment of hepatitis C virus infection. ACS Nano. 2012;6(11):9522-31