Introduction: As current treatments for chronic neck or low back pain are not optimal, one new approach being explored is the replacement of a degenerated intervertebral disc (IVD) with a bioengineered IVD. However, bioengineering the Multilamellated annulus fibrosus has been problematic as it is composed of outer annulus fibrosus (OAF) and inner annulus fibrosus (IAF) which differ in their composition. It has been previously shown that when these cells were cultured on a single layer of aligned nanofibrous polycarbonate urethane (PU-ADO) membranes, they retain partially their differentiated phenotypes by accumulating different extracellular matrix molecules, such as type II collagen, which is exclusively accumulated by IAF tissues. In this study, we examine whether Multilamellated OAF and IAF tissues can be generated in vitro.
Methods: Multilamellated constructs were generated by seeding 3 layers of PU-ADO scaffolds with OAF and IAF cells in spinner flasks, cultured up to two weeks. Harvested tissues were analyzed by biochemical assays for DNA content, proteoglycan content, collagen content, as well as histological analysis.
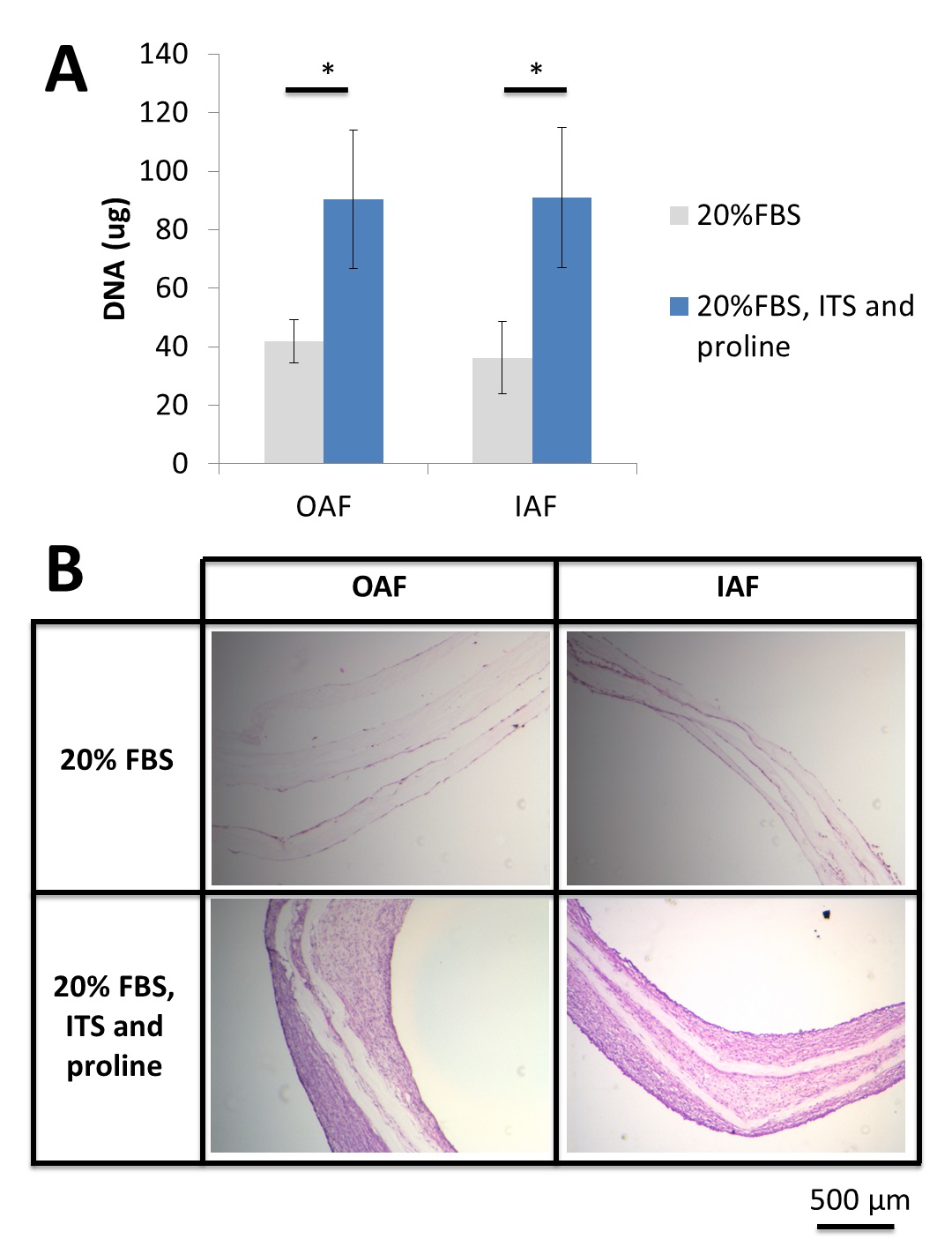
Results and Discussion: The previously employed medium condition – DMEM containing 20% FBS - was ineffective in yielding tissue reproducibly in the multilamellated PU-ADO constructs by either OAF or IAF cells.
Supplementation of the serum-containing media with insulin-transferrin-selenium (ITS+) and proline improved the robustness of formed OAF and IAF tissues in the multilamellated PU-ADO constructs after 2 weeks in culture by increasing tissue cellularity.
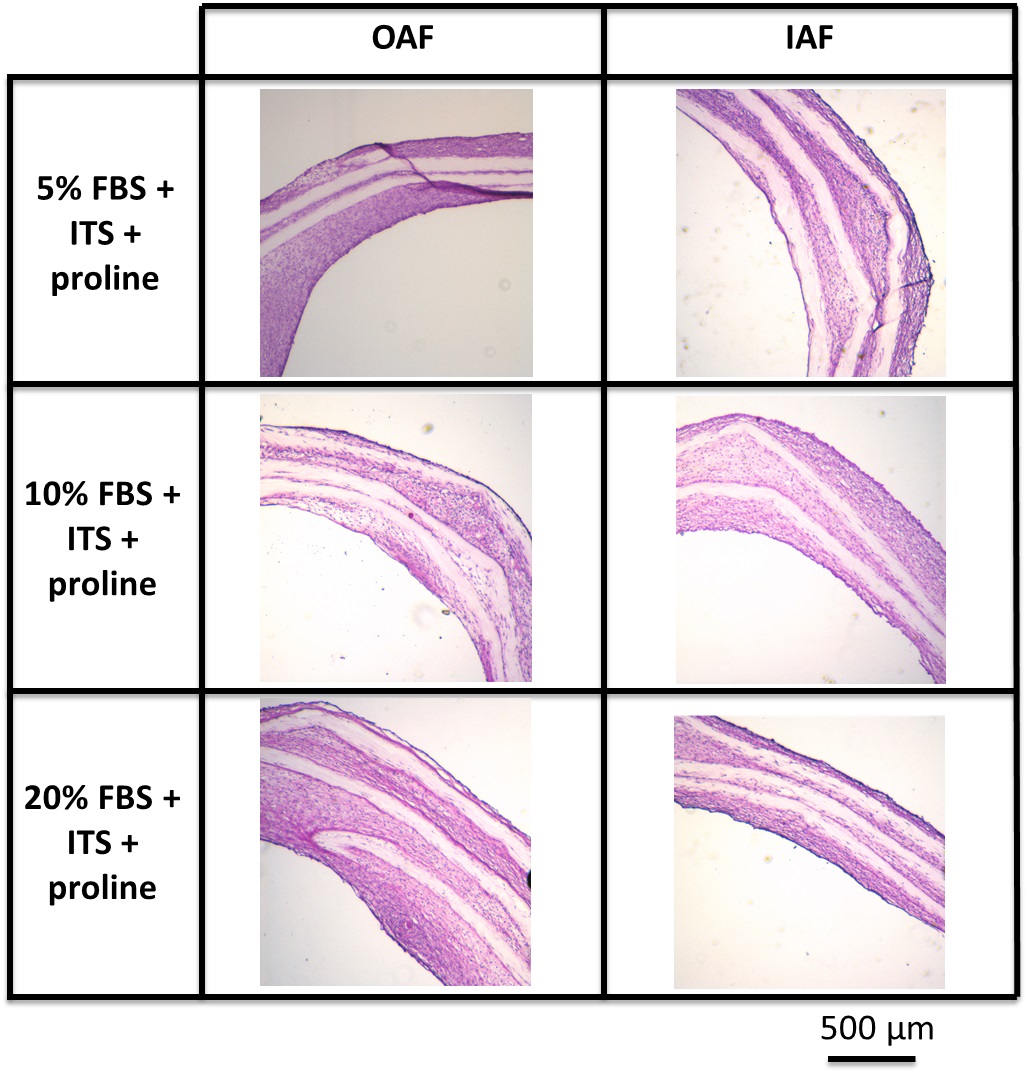
Reduction of serum concentration in medium supplemented with ITS and proline did not affect the increase in cellularity of the tissues formed in vitro. The improvement in the robustness OAF and IAF tissues was a result of an increase in cell proliferation promoted by ITS+ over the culture period.
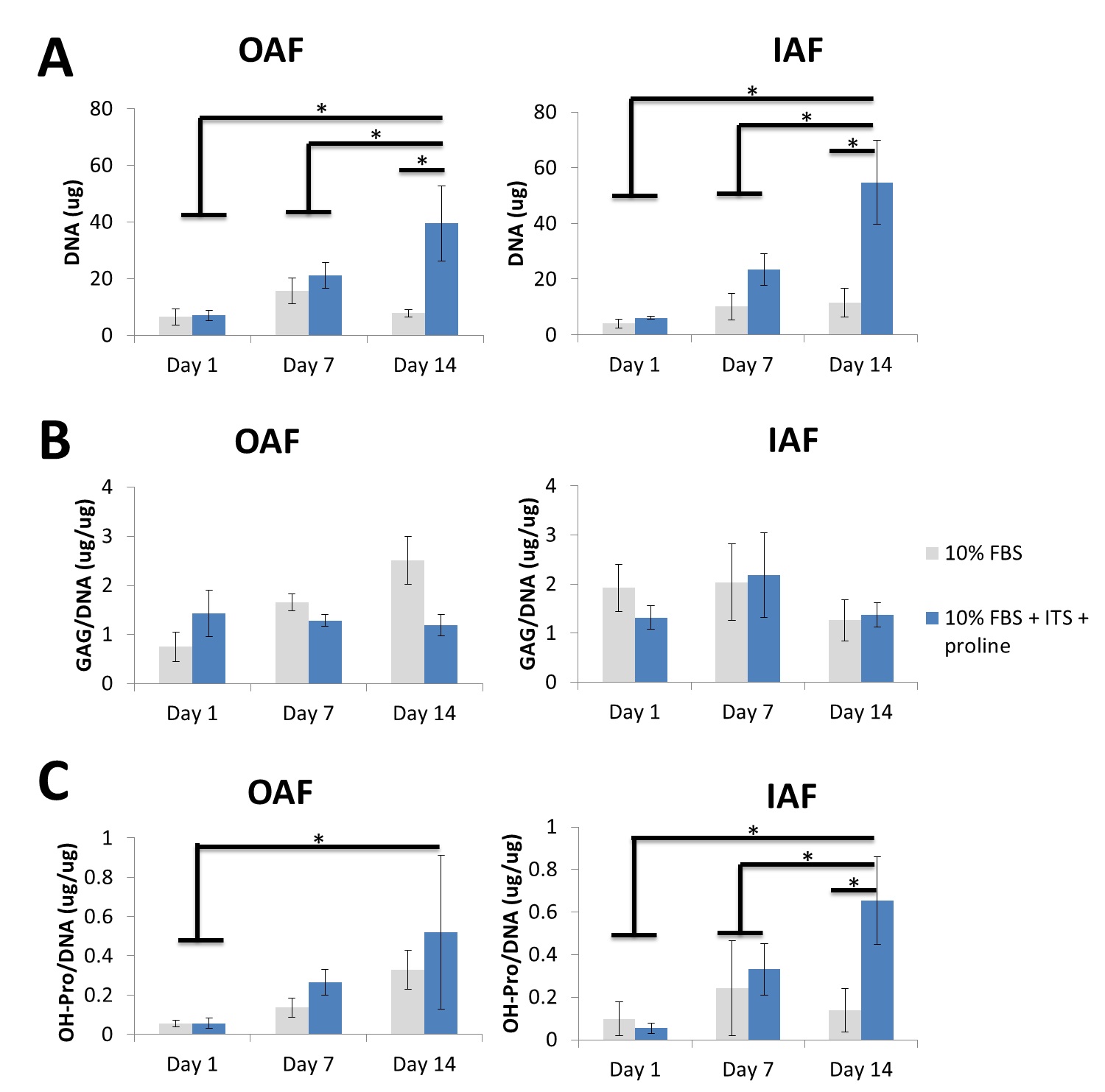
While ITS and proline supplementation promoted robustness of OAF and IAF tissues in the multilamellated PU-ADO constructs, IAF tissues did not retain the differenated phenotypes as indicated by the inability to accumulate type II collagen - a characteristic extracellular matrix molecules for IAF tissues.
Conclusion: Further culture conditions are required and currently on going to identify the in vitro conditions that favour formation of IAF and OAF tissues, such as the application of compressive loading and tensile strains that resembles their native mechanical environment.