Introduction: Textile technologies have opened a new area in tissue engineering. Precise control over the distribution of different cell types and microstructure of fabricated constructs are considered as key advantages of textile techniques. Insufficient mechanical properties of cell-carrying fibers made of hydrogels have limited their usage in textile processes. Thus, the concept of composite living fibers (CLFs) that can withstand textile processing has been recently introduced to address this challenge. However, these CLFs could not support long term cellular viability and functionality. Here we introduced the hybrid bioactive hydrogels for fabrication of the new generation of CLF that not only could support cellular attachment and provide a suitable environment for cell proliferation and viability of anchoring cells but also can accurately control cellular alignment.
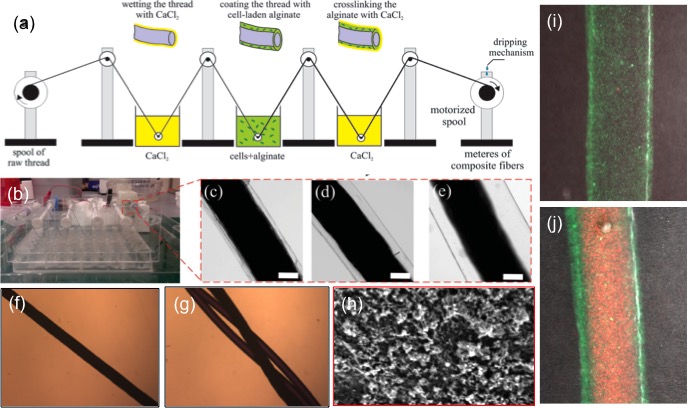
Materials and Methods: CLFs were fabricated by coating a flexible biocompatible and mechanically strong collagen-based fiber by a cell-laden hydrogel layer from alginate and gelatin methacrylate (GelMA) hybrid with concentrations of 0.5-2% and 5-7.5% w/v, respectively. A motorized spool drew the core fiber through a mixed solution of alginate, GelMA, and cells and then through a solution of 2% (w/v) CaCl2 to crosslink the alginate part and form a polymeric network template (Fig. 1a). GelMA part was crosslinked by UV irradiation (365 nm, 850 mW). A dripping mechanism is designed to wash the excessive calcium from the composite fibers while keeping them in a wet environment by using tris-buffered saline (TBS). Alginate could also be removed by a calcium chelator to leave a hydrogel layer from GelMA (Fig. 1h). Fibroblast viability and metabolic activity were assessed by a standard live/dead assay and PrestoBlue assay. Cellular morphology and alignment were monitored by immunostaining of α-actin and nuclei.
Results and Discussion: CLFs containing fibroblasts were created and the thickness of the gel layer could be changed from 20µm to 600µm by adjusting the drawing speed and the prepolymer concentrations (Fig. 1b-e). The encapsulated 3T3 fibroblasts showed high cellular viability (data not shown). In addition, the metabolic activity of the encapsulated cells was assessed and showed a continuous increase over 7 days of culture. Immunostaining results confirmed normal cellular morphology and alignment of encapsulated cells along fiber’s axis (Fig. 1i,j).
Conclusions: A scalable technology for creating mechanically strong cell-laden fibers is reported that allows continuous fabrication of CLFS in reel-to-reel format. The fabricated fibers supported viability and proliferation and alignment of fibroblasts.
National Science Foundation (EFRI-1240443); Office of Naval Research Young National Investigator Award; National Institutes of Health (HL092836, DE019024, EB012597, AR057837, DE021468, HL099073, EB008392); MIT-Italy program (ProgettoRocca); Polimi International Fellowship (PIF)