Introduction: Bioactive ceramics used in bone replacement and regeneration are generally of a certain degree of reactivity and thus able to release bioactive ions. However, high reactivity leads to a burst release of their compositional ions, resulting in an excessive dosage and an undesired large fluctuation of pH from the physiological level caused by the released alkaline-earth metal ions (e.g. Ca or Sr etc.). This is a general concern for most of the degradable bioinorganic materials. Therefore, it is crucial to control the initial interfacial chemical interaction to avoid the burst release of ion. To this end, in this study, we used a simple solution-based method to convert the surface highly reactive phase to less reactive biomorphic crystals. Notably, the newly formed crystals have a well-organized shape full of micro/nanotopographic features, acting as a bioactivity enhancer.
Materials and Methods: Strontium zinc silicate (SZnS) biomedical coatings were deposited on biomedical grade Ti alloy plates (Ti-6Al-4V, Baoji Junhang Metal Material Co., Ltd.) using an atmosphere plasma spraying system (APS, Sulzer Metco, Switzerland).
To investigate the surface reactivity, the coating samples were incubated in cell culture medium with/without 10%FBS at 37oC for 5 hours without extra CO2 supplementation. To take the advantage of the surface reactivity of the SZnS coating, an interfacial intervention was used to accelerate the interfacial reaction. Briefly, coating samples were put in a small glass bottle with a volume of 10ml containing 3ml buffers containing 4.2mM CO32-, then subjected to autoclave sterilization. For comparison, some coating samples subjected to UV sterilization for 1h were used as control samples.
SEM, XRD, TEM and Raman spectroscopy were used to characterize the physical structure and crystalline structure of the coating. Preosteoblasts (MC3T3-E1) were used to evaluate the cytocompatibility of the coatings before and after interfacial intervention. The cell adhesion, collagen production and ALP activity were investigated.
Results and Discussion: Figure 1 displays the surface morphology of the coatings after incubation in cell culture medium with/without FBS. From Figure 1a, it can be seen that foxtail-like SrCO3 crystals are formed on the surface. With a closer observation, we find that the crystals are built up from fibrous crystallites with 50nm in thickness, which is a characteristic of silica-carbonate biomorphs (Figure 1a). In the case of incubation in the serum-free medium, some flower-like crystals grow on the coating surface (Figure 1b). Preminent differences in the surface morphology are observed for the coating after autoclave sterilization (Figure 1c-f). The newly formed crystal assemblies nearly cover the whole coating surface. The formation of these interweaving crystals is driven by the interfacial reaction of the “reactive” substrate. Cell experiments show that the coating surface with biomorphic SrCO3 crystals enhances the adhesion of osteoblasts and upregulates the ALP activity and collagen production in the osteoblasts.
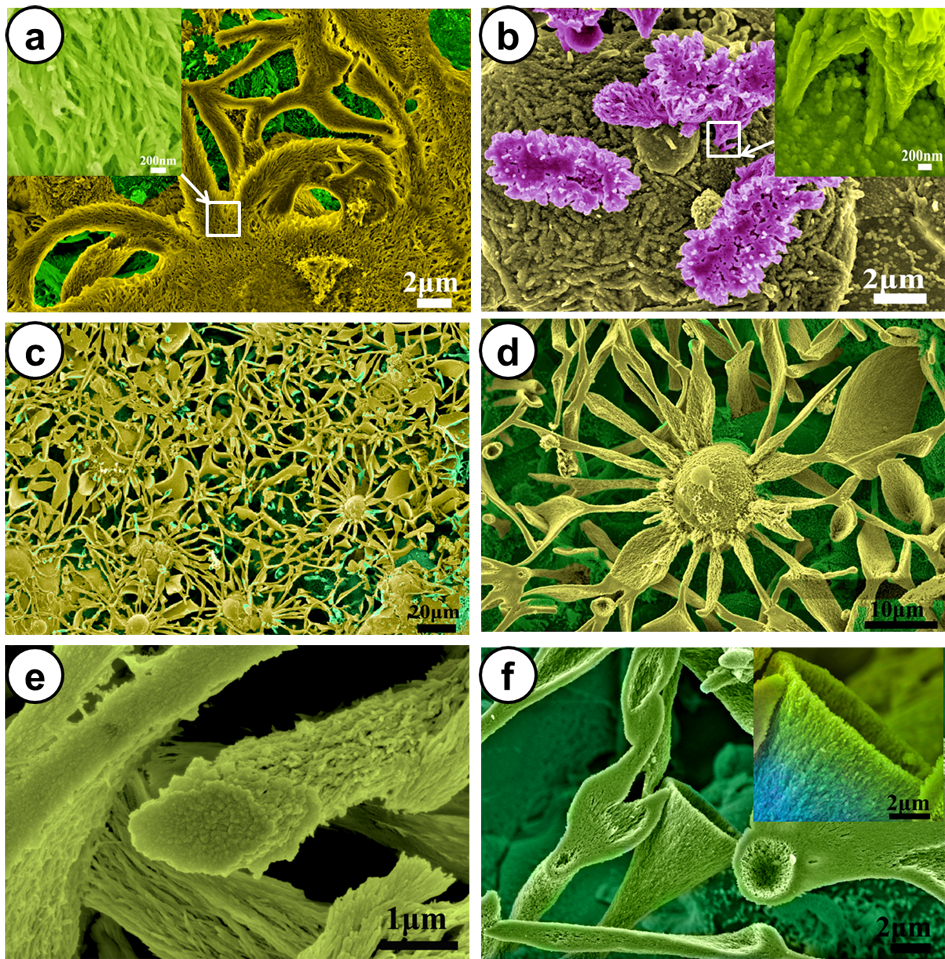
Conclusion: We report interfacial reaction-driven formation of silica-carbonate biomorphic crystals with hierachical micro/nanostructures. The formation of the biomorphic crystals enhanced the adhesion of osteoblasts and upregulate the ALP activity and collagen production in the osteoblasts.
Authors want to thank the Shenzhen Peacock Innovation Team (110811003586331) and Shenzhen Key Laboratory of Marine Biomedical Materials (ZDSY20130401165820356) for their support.