Introduction: Stem cell-based therapy hold great promise for regenerating lost tissues such as bone, and synthetic grafting biomaterials, such as hydrogels, have been developed to provide structural support and promote desirable cell fates and tissue formation. However, the efficacy of previously developed scaffolds for bone repair are often limited by poor cell survival, non-uniform cell distribution, or the lack of macroporosity that is desirable to promote cell migration and new matrix deposition. To overcome the above limitations, we recently developed microribbon (μRB)-like, crosslinkable elastomers as scaffold building blocks, which can encapsulate cells in 3D while simultaneously forming a highly macroporous scaffold[1]. Our previous studies showed that μRBs facilitate uniform cell encapsulation, allow formation of 3D scaffolds with tunable macroporosity, and support rapid cell spreading and proliferation in vitro. We hypothesized that macroporous μRB-based scaffolds would promote stem cell engraftment and survival after transplantation, and enhance host tissue ingrowth and promote bone regeneration. The goal of the current study was to evaluate the potential of gelatin-based μRB scaffolds for in vivo bone repair using a critical-sized, mouse cranial defect model.
Materials and Methods: Photocrosslinkable gelatin mRBs were synthesized by wet-spinning as we previously reported[1]. Prior to use, mRBs were rehydrated in phosphate-buffered saline and mixed with trypsinized GFP/Luc+ mouse adipose-derived stem cells (ADSCs). To fabricate macroporous scaffolds, molded mRBs were photocrosslinked (365 nm, 5 min, 4 mW/cm2) in the presence of a photoinitiator. The resulting scaffolds were characterized by unconfined compression testing and scanning electron microscopy for morphology. The stiffness of individual μRBs was measured using atomic force microscopy. To compare the efficacy of mRB-based scaffolds for supporting bone repair with that of existing hydrogel platforms, hydrogels of methacrylated gelatin were used as control groups. To assess the ability of mRB-based scaffolds to promote ADSC survival and bone regeneration in vivo, mouse critical sized cranial defects (4 mm in diameter) were treated with the following five groups including: (1) μRB+ADSC, (2) hydrogel (HG) +ADSC, (3) μRB only, and (4) HG only, (5) untreated defect (neg ctrl). All groups were monitored for up to 6 weeks, and outcomes were evaluated using bioluminescence imaging, micro-CT, and histology.
Results and Discussion: Photocrosslinkable gelatin-based μRBs were synthesized by wet-spinning as previously reported[1] (Fig. 1). The resulting μRBs are 40-70 m in width and injectable through 16G needle, and allows direct encapsulation of ADSCs in 3D (Fig. 1). Upon photocrosslinking, the microribbons formed a highly macroporous scaffold that supported cell spreading and proliferation (Fig. 1). Results from a critical-sized, mouse cranial defect model demonstrated that microribbon-based scaffolds significantly enhanced the survival and proliferation of transplanted adipose-derived stromal cells compared with conventional hydrogels (Fig. 2). Macroporosity among microribbons facilitated mineralized bone formation (as shown by microCT) and host tissue ingrowth (histology) (Fig. 3). The observed enhancement of cell survival and proliferation further promoted the paracrine-signaling effects of adipose-derived stromal cells for stimulating endogenous bone regeneration.
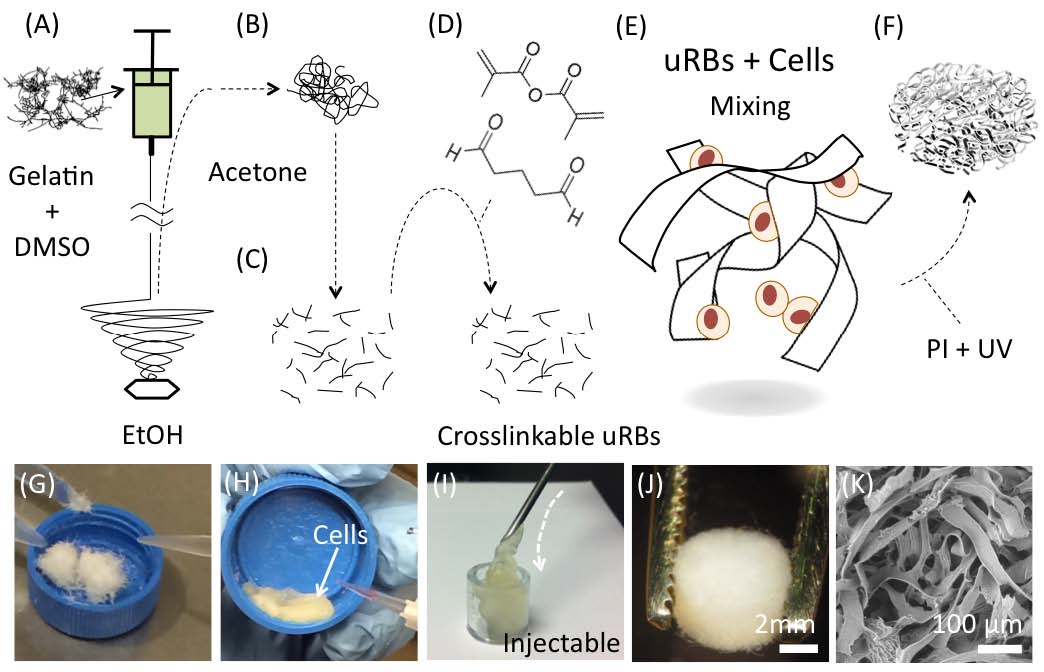
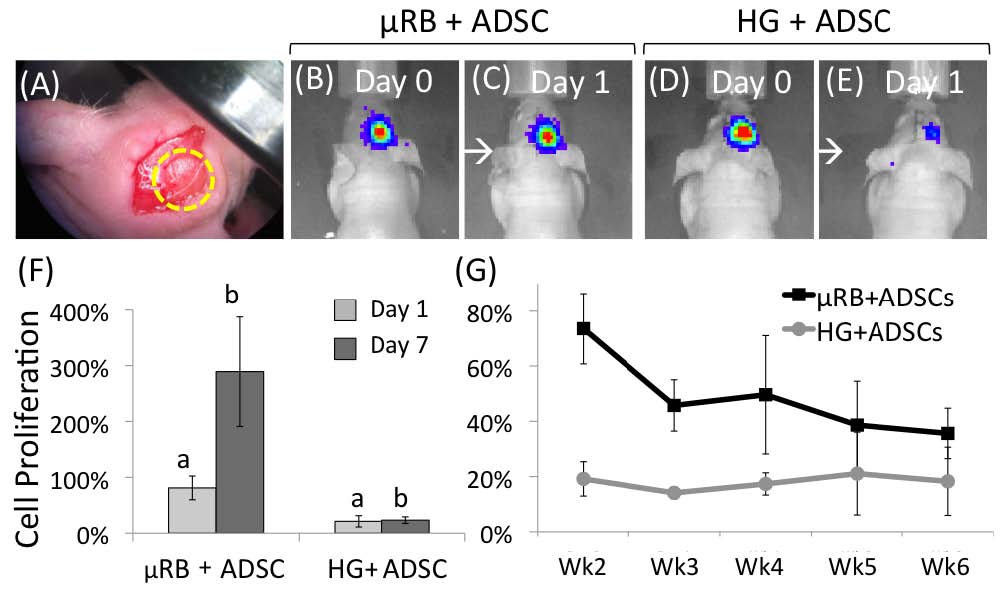
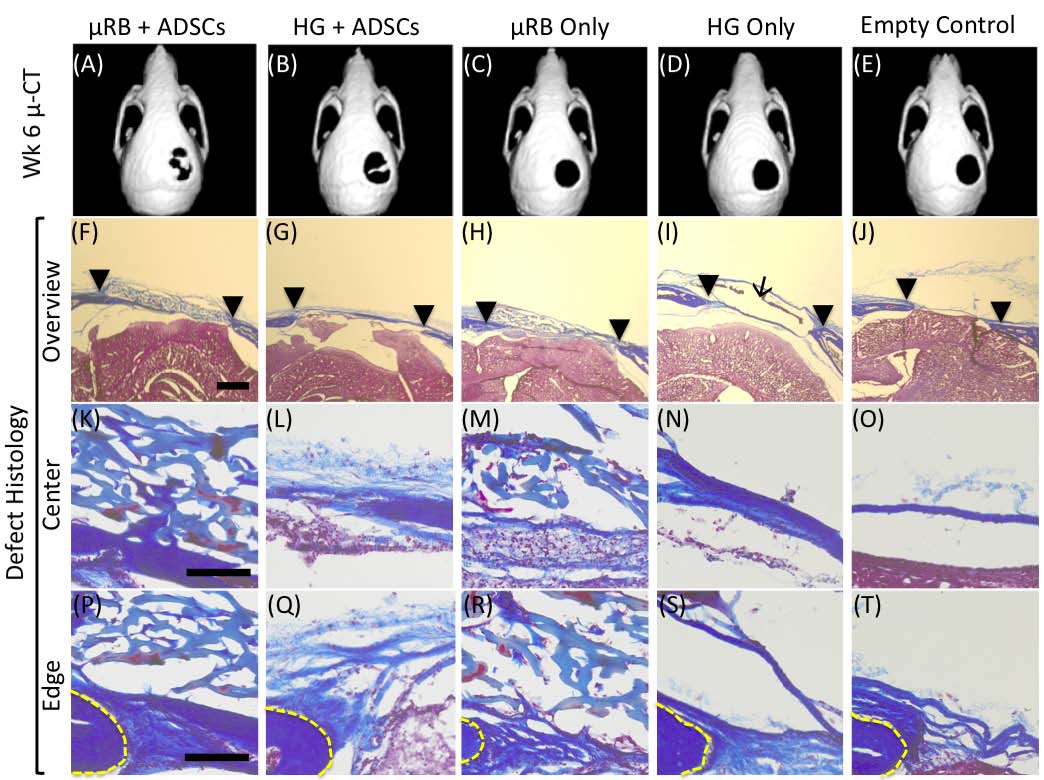
Conclusions: Here we validate the efficacy of photocrosslinkable μRBs as injectable macroporous scaffolds for stem cell delivery and tissue regeneration. Compared to conventional nanoporous hydrogels, our μRB hydrogels are macroporous, which led to enhanced stem cell survival and engraftment, accelerated vascularization and endogenous bone repair. We anticipate such microribbon-based scaffolds will provide a novel injectable matrices for stem cell-based therapies to enhance tissue regeneration outcomes by substantially improving cell survival, vascularization and new extracellular matrix deposition.
The authors would like to acknowledge the following funding support including NIH R01DE024772 (F.Y), California Institute for Regenerative Medicine (Grant #TR3-05569) (F. Y.), National Science Foundation CAREER award(CBET-1351289, F.Y.) Stanford Chem-H Institute Biomaterials Seed Grant (F. Y.), Stanford Coulter Translational Grant, and Stanford Child Health Research Institute Faculty Scholar Award (F. Y.)
References:
[1] Han L-H, Yu S, Wang T, Behn AW, Yang F. Microribbon-Like Elastomers for Fabricating Macroporous and Highly Flexible Scaffolds that Support Cell Proliferation in 3D. Adv Funct Mater. 2013;23:346-58.