Introduction: Mechano growth factor (MGF) and its C-terminal E-peptide with 24 amino acids, MGF-Ct24E, have superiority in resolving the delayed or failed bone repair derived from shortness of suitable biomechanical stimulation [1],[2]. The purpose of the present study is to verify the feasibility of utilizing chitosan (CS) as a carrier for sustained delivery of MGF-based growth factor.
Materials and methods: The CS microspheres encapsulated with MGF-Ct24E are prepared using emulsion-ionic cross-linking method in the presence of tripolyphosphate (TPP) and the CS/MGF-Ct24E microspheres are obtained.
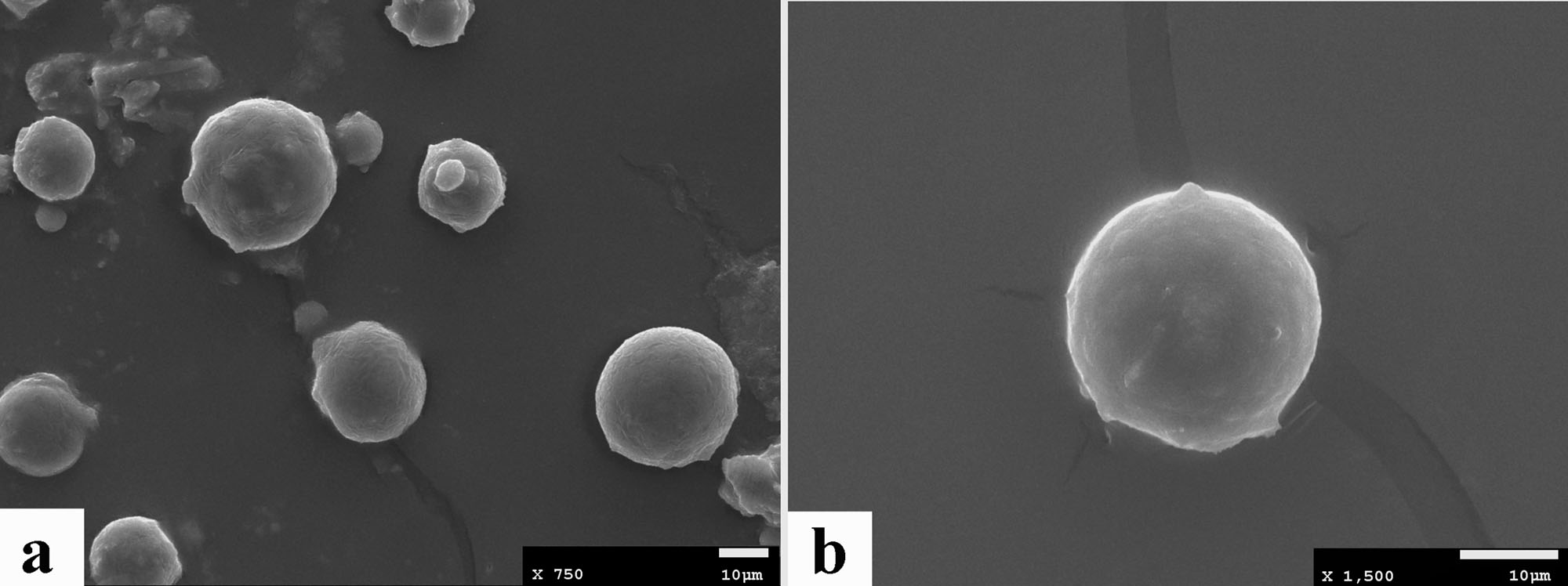
Results and discussion: The microspheres are micron-sized and spherical in shape with smooth surface morphology.
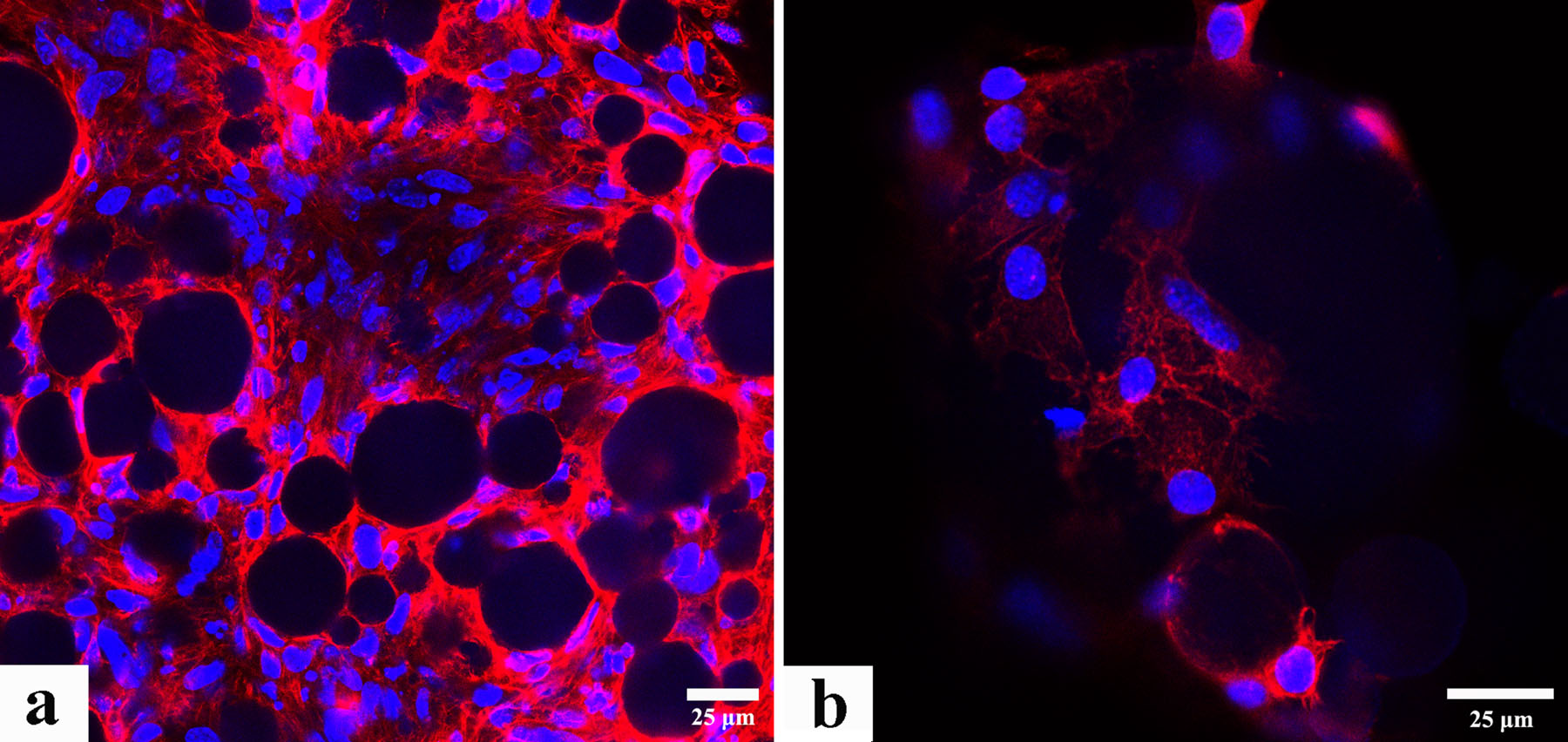
The TPP component disintegrates in advance of CS matrix and the MGF-Ct24E maintains sustained delivery during in vitro hydrolytic degradation. With the disappearance of TPP, the total weight loss of CS/MGF-Ct24E is 32% and the release amount reaches 84.6% after degrading for 2 weeks. In vitro bioactivity assays reveal that the MGF-Ct24E can accelerate MC3T3-E1 cells proliferation and delay their differentiation as well. The encapsulated MGF-Ct24E shows long-term effects after being loaded in the CS microspheres and the cells exhibit excellent morphology on the surface of microspheres.
Conclusions: The continuous delivery of MGF-Ct24E provides a new perspective on resolving the unsatisfactory bone reconstruction associated with stress shielding.
This research was financially supported by the National Natural Science Foundation of China (Nos. 11272038, 31470915, 11421202, and 11032012), and the Pearl River S&T Nova Program of Guangzhou, China.
References:
[1] Luo ZW, Jiang L, Xu Y, et al. Mechano growth factor (MGF) and transforming growth factor (TGF)-b3 functionalized silk scaffolds enhance articular hyaline cartilage regeneration in rabbit model. Biomaterials, 2015, 52: 463-475.
[2] Goldspink G. Mechanical signals, IGF-I gene splicing, and muscle adaptation. Physiology, 2005, 20:232-238.