Introduction: Despite the use of PEGylation to improve in vivo stability of traditional polymeric siRNA delivery vehicles, circulation half-lives remain short (~20 min) [1]. Serum opsonins and complement proteins adsorb onto these particles, decreasing stability and increasing recognition and uptake by the mononuclear phagocyte system (MPS)[2]-[4]. To improve intravenous polyplex delivery of RNAi therapeutics, it is imperative to design nanocarriers with improved resistance to the challenges of a complex protein environment. The current work compares zwitterionic corona chemistry to traditional linear and branched PEG-based architectures for predictors of best in vivo function. To our knowledge, this work describes and provides initial characterization of the first endosomolytic zwitterion-based polymeric siRNA delivery system.
Materials and Methods: Poly[(ethylene glycol)-b-[(2-dimethylamino)ethyl methacrylate) (DMAEMA)-co-(butyl methacrylate) (BMA)] (PEG-DB), poly[(ethyl ether methacrylate) (POEGMA)-b-p(DMAEMA-co-BMA)] (POEGMA-DB), and poly(methacryloyloxyethylphosphorylcholine (PMPC)-b-p(DMAEMA-co-BMA)] (PMPC-DB), were RAFT polymerized with an optimized 50:50 ratio of DMAEMA and BMA[5]. Polymers were characterized with GPC and 1H-NMR. Polyplexes formulated from these polymers and siRNA were characterized with DLS and TEM. These polyplexes were assessed for uptake and in vitro knockdown of the model gene luciferase in breast cancer cells (MDA-MB-231), as well as for endosomal escape function and cytotoxicity. Finally, isothermal titration calorimetry was used to probe adsorption of bovine serum albumin.
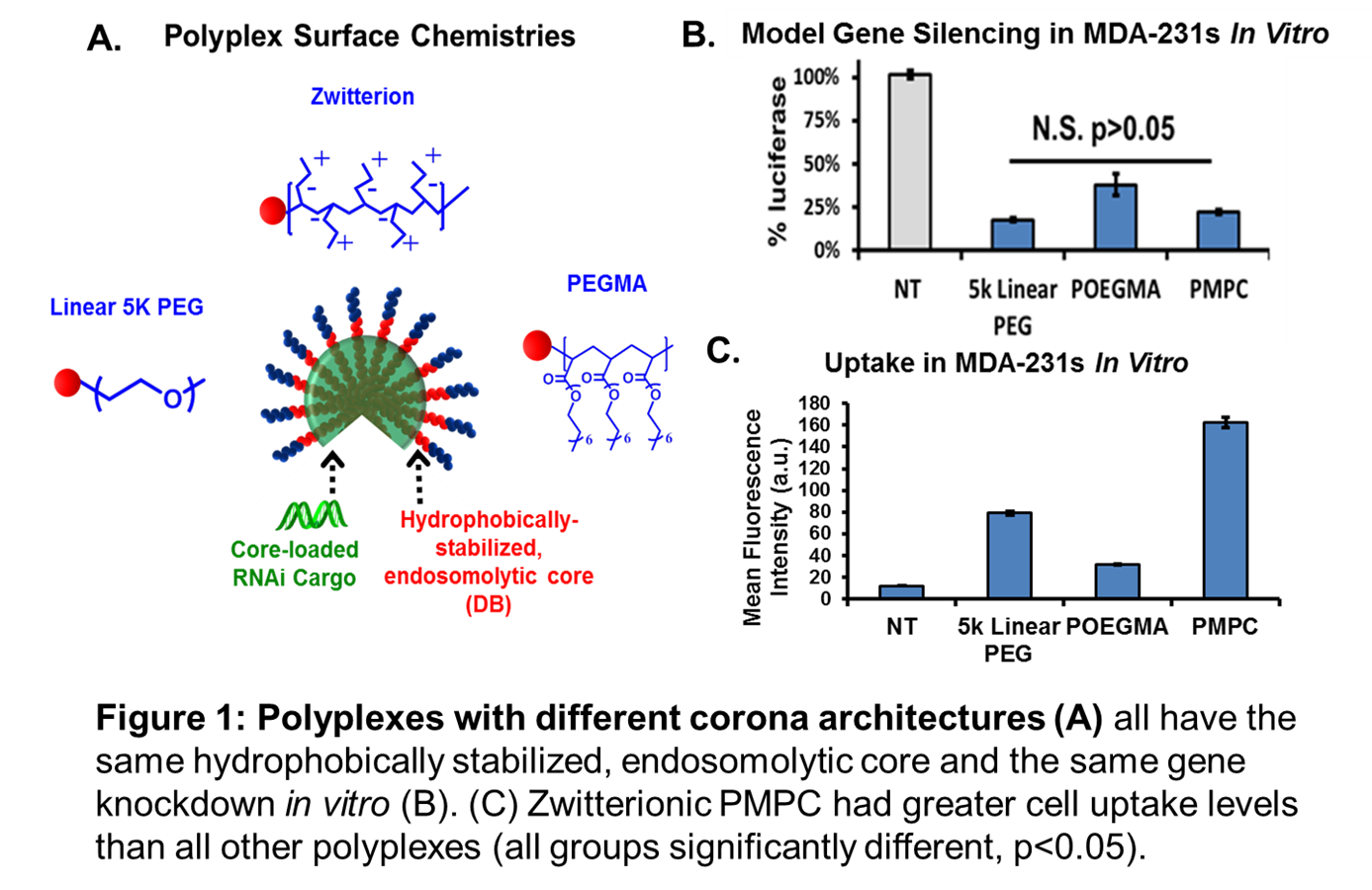
Results and Discussion: Overall, zwitterionic PMPC-DB notably outperformed PEG-DB and POEGMA-DB in terms of increased cell uptake (Fig 1C) and reduced protein adsorption (Fig 2). Each polymer formed siRNA polyplexes of similar size (~150 nm) and had neutral zeta potential with low polydispersities. All polyplexes exhibit similar knockdown of the model gene luciferase (Fig 1B) and equally low levels of toxicity.
Based on isothermal titration calorimetry (Fig 2), zwitterionic PMPC-DB was least favorable to protein adsorption compared to PEG-DB, POEGMA-DB, and a control formulation with a purely cationic DMAEMA corona (D-DPB), as evidenced by its large change in Gibbs free energy (ΔG). However, PMPC-DB had the greatest increase in enthalpy, suggesting that more energy is required to displace the hydration shell of zwitterionic polyplex coatings than for traditional PEG coatings. While the displacement of cage-like hydration shells resulted in favorable entropy changes for PMPC-DB, this effect was outweighed by the enthalpy required to displace them, resulting in higher ΔG values.
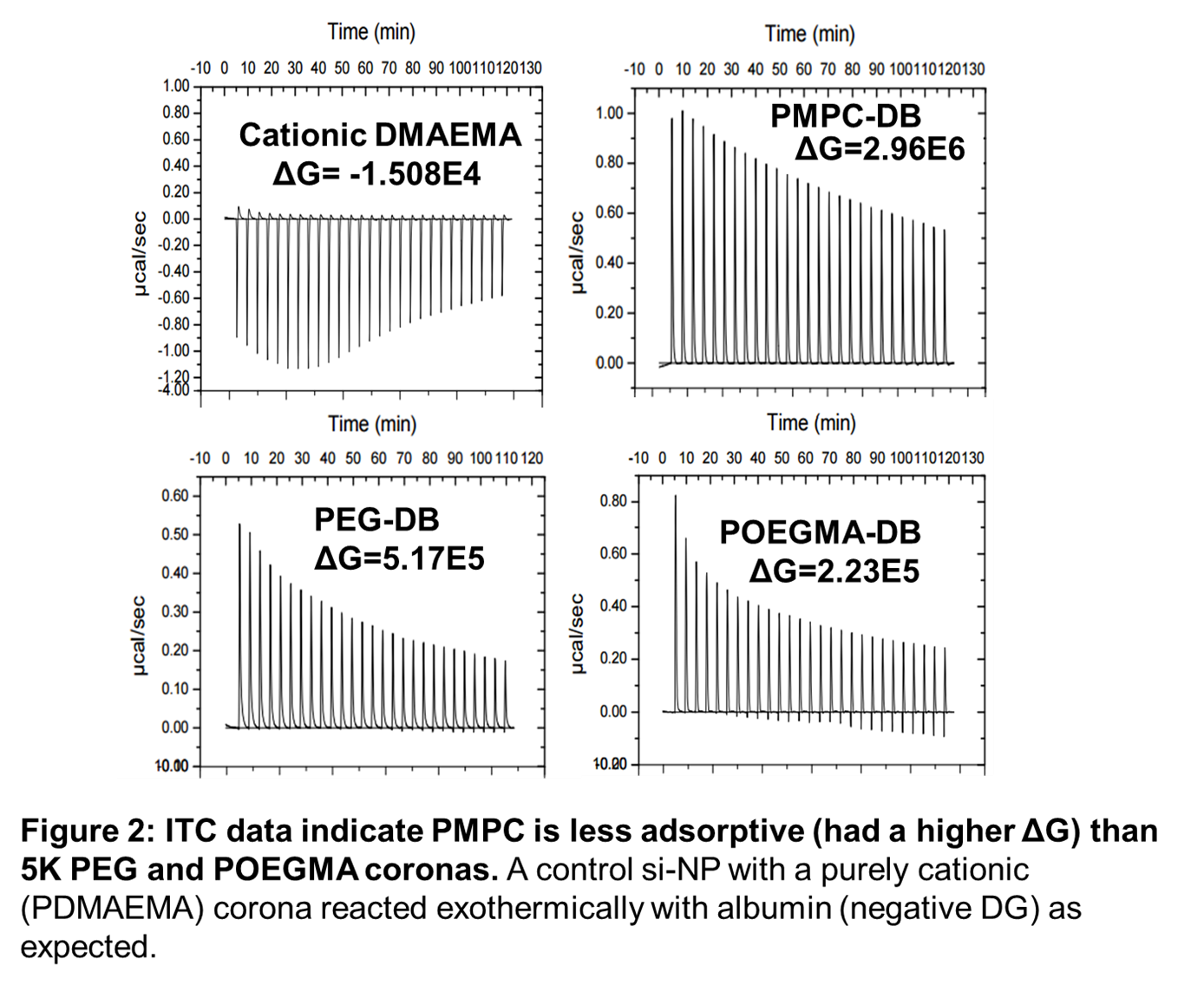
Conclusion: In conclusion, novel zwitterionic siRNA polyplex coronas show promise for improved in vivo pharmacokinetics relative to conventional PEG based on increased uptake and reduced adsorption of serum proteins. This is a significant finding as there is a known direct correlation between level of opsonization and rapidity of hepatic uptake of IV-injected nanocarriers [4]. In vivo pharmacokinetic effects of the PMPC corona will be compared to PEG in ongoing studies.
This work was supported in part by National Science Foundation Graduate Research Fellowship Grant No. 1445197.; We would like to thank members of the Duvall and Giorgio labs for scientific discussions and support regarding this work.; We would like to thank Dr. Joel Tellinghuisen for his guidance regarding isothermal titration calorimetry methods.; This work was supported in part by the NIH through the grant 1R01EB019409.
References:
[1] Miteva M, Kirkbride KC, Kilchrist KV, Werfel TA, Li H, Nelson CE, Gupta MK, Giorgio TD, Duvall CL. "Tuning pegylation of mixed micelles to overcome intracellular and systemic sirna delivery barriers." Biomaterials. 2015;38:97-107
[2] Pelaz B, del Pino P, Maffre P, Hartmann R, Gallego M, Rivera-Fernández S, de la Fuente JM, Nienhaus GU, Parak WJ. "Surface functionalization of nanoparticles with polyethylene glycol: Effects on protein adsorption and cellular uptake." ACS Nano. 2015;9:6996-7008
[3] Owens DE, Peppas NA. "Opsonization, biodistribution, and pharmacokinetics of polymeric nanoparticles." International Journal of Pharmaceutics. 2006;307:93-102
[4] Nagayama S, Ogawara K-i, Fukuoka Y, Higaki K, Kimura T. "Time-dependent changes in opsonin amount associated on nanoparticles alter their hepatic uptake characteristics." Int J Pharm. 2007;342:215-221
[5] Nelson CE, Kintzing JR, Hanna A, Shannon JM, Gupta MK, Duvall CL. "Balancing cationic and hydrophobic content of pegylated sirna polyplexes enhances endosome escape, stability, blood circulation time, and bioactivity in vivo." ACS Nano. 2013;7:8870-8880