Introduction and Motivation: In recent years, magnesium alloy is used to be a kind of medical biodegradable material. Magnesium is not only an essential element in the body, but also the element which can be excreted by the kidneys efficiently.
However, magnesium alloy corrodes so quickly that it may cause surface of implant would releases hydrogen and pH value. And then it could cause harm to cells. On the other hand, the magnesium alloy implants corrode too fast to maintain the function of implant until the healing of tissue.
Therefore, much recent research about magnesium alloy has focused on controlling the corrosion rate. In our research, it will be to study the degradation of magnesium alloy in saline solution. The experimental results will be compared with the findings from the mathematical model of dynamic changes of magnesium alloy corrosion.
Material and Method: The in vitro corrosion behavior of magnesium alloys is affected by many factors[1]. In the experiment of this study, we will study on the influence of chloride concentration and composition of magnesium alloys on the corrosion behavior by the Micro-CT (micro computed tomography).
In the first step, we make the guiding plates for specimens of magnesium alloy by rapid prototyping. The guiding plates are able to be the standard of specimen degradation, we can use it to make sure the position of specimens in the CT image and simplify the conditions of degradation.
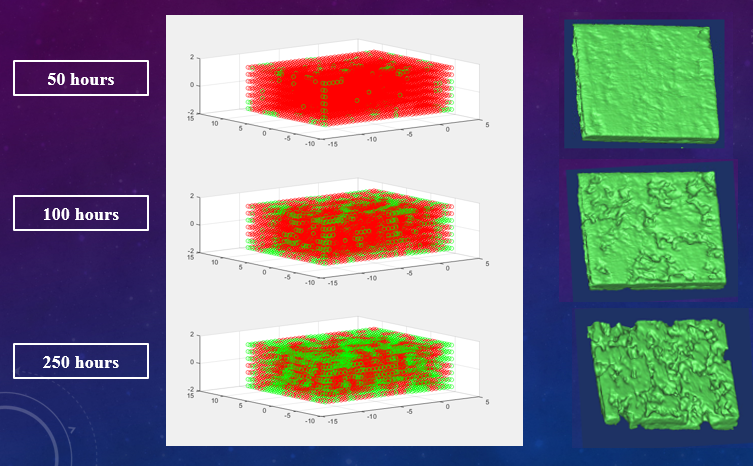
In the next step, we use the physiological saline solution and deionized water to combination the solution with different chloride concentration. And then we put the specimens into the solution for the corrosion test. The CT image, surface photographs and weigh are measured on every 50 hours, and the experimental results will be compared with the finite element model.
Result: In the primary results of the corrosion test, we make sure that CT image can be a way to quantify the corrosion behavior of magnesium alloy. We will deal with the raw data into Mass Loss (ML),corrosion morphology and corrosion rate in the local and general site by CT image in the near future.

On the other hand, in the primary results of the computer simulation, the results of the corrosion morphology by the simulation is approximately identical with corrosion test.
As a biodegradable implant for the area with stagnating body fluid flow in the human body, magnesium alloy can cause the increase of local pH values and release the hydrogen. Those may damage the human cell[2]. Therefore, how to control the rate of degradation of magnesium alloys become very important. The purpose of this study is finding out the mathematical model equation of each factors and corrosion rate. After that we will try to find the ways to overcome the limitations of medical magnesium alloy.
References:
[1] Yuki Soya, Shoichiro Yoshihara, Yuki Ohmura, Bryan J. MacDonald and Emmet Galvin, Corrosion Behavior of Engineering Materials in Flow Field , Advanced Materials Research Vol. 922 (2014) pp 722-727,2014.
[2] CHI-HUA CHU, The Improvement of Surface Roughness and Corrosion Resistance for Biodegradable AZ61 Magnesium Alloy, U0026-2911201314103300, 2013.