Introduction: An ideal tissue engineering scaffold (TES) should have an architecture that facilitates tissue formation. They should be non-immunogenic in nature to prevent any inflammatory reactions and should be able to resorb at a controlled rate. Their surface topography should enhance cell adhesion and proliferation[1]. Polymer fibres due to their high surface area have emerged as a new class of biomaterials for tissue engineering applications. In this project, we have synthesized porous, aligned PHA fibres using pressurized gyration process (PGP). It is a robust, cost-effective and a novel technique that generates fibres of desired properties[2]. PHA, which is the chosen polymer for this study, is non-immunogenic, hydrophobic storage polymers produced by a variety of bacterial species under nutrient-limiting conditions. They are biodegradable in nature and exhibit thermoplastic properties[3]. This study describes the production of PHAs, synthesis of aligned porous PHA fibres using PGP and their evaluation as TES.
Experimental Methods: Production and characterisation of PHAs: PHAs were produced by bacterial fermentation of Cupriavidus necator in bioreactors using edible oil as the carbon source. The PHA produced was extracted, purified and characterised using Gas chromatography-Mass Spectrometry (GC-MS) and Nuclear magnetic Resonance (NMR). Synthesis and characterisation of PHA fibres: Purified PHA was used to generate aligned fibres by pressurized gyration as described in Fig1.
Figure 1: Experimental set up of the pressurised gyration process.
PHA fibres were viewed under the scanning electron microscope (SEM) to determine their size and surface topography. In vitro biocompatibility studies C2C12 myoblast cells and Human microvascular endothelial cells (HMEC-1) were seeded on these fibres for 1, 3 and 7 days. Cell viability studies were carried out using MTT assay. Confocal imaging and SEM studies were carried out after each time point. 5wt% PHA films were used in all experiments for comparison.
Results and Discussion: Production of P(3HB), P(3HB) aligned fibres and their characterisation: PHA was successfully produced, extracted and purified. GC-MS and NMR data confirmed that the PHA produced was poly(3-hydroxybutyrate) or P(3HB). Aligned P(3HB) fibres were generated by pressurized gyration as shown in Fig 2. SEM images revealed porous fibres of 9±2.6 mm diameter.

Figure 2: SEM micrograph of the porous aligned P(3HB) fibres
Cell proliferation studies: Cell attachment and proliferation (both HMEC-1 and C2C12) was found to be highest on the aligned fibres compared to the P(3HB) film and tissue culture plastic which was used as the positive control as represented in Fig 3(a-b). SEM and the confocal images further confirmed that the aligned P(3HB) fibres supported the adhesion and proliferation of both HMEC-1 and C2C12 cells (Fig 3c-3f). HMEC-1 cells grew in an aligned manner on the gyrospun P(3HB) fibres as shown in Fig 3d.
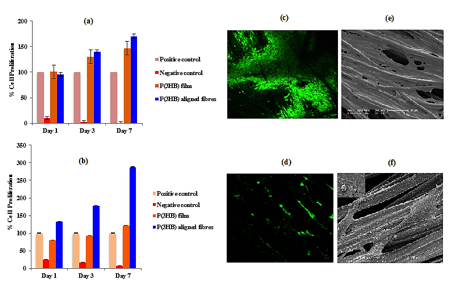
Figure 3: Cell proliferation data of the (a) C2C12 cells and (b) HMEC-1 cells on the P(3HB) aligned fibres and P(3HB) film. Confocal images of the (c) C2C12 cells and (d) HMEC-1 cells and the SEM images of the (e) C2C12 cells and (f) HMEC-1 cells on the P(3HB) aligned fibres.
CONCLUSION: Cell adhesion, proliferation and alignment were significantly higher on the aligned P(3HB) fibres compared to the P(3HB) film and hence, proved to be an ideal biomaterial for TE applications.
We would like to thank University of Westminster and University College London for their support
References:
[1] Hutmacher D. W., Biomat 24:2529-2543, 2000
[2] Mahalingam S. et al., Macromolecular rapid Communications 14: 1134-1139, 2013
[3] Basnett P. et al., Adv. Eng. materials 6: 330-343, 2012