Introduction: Flexible electronics are ideally suited for brain-machine interfaces because they are mechanically compliant and can match the mechanical properties of many types of excitable tissue in the brain and peripheral nerve. Peripheral nerve interfaces (PNI) with penetrating electrodes can record and stimulate with higher information bandwidth, but are subject to host-biomaterials responses, which limit the effective electrode resolutions. Surface electrode systems permit minimally-invasive modes for neural interfaces by neural therapy by directly interfacing with tissue surfaces to minimize inflammation and improve reliability during recording and stimulation. Furthermore, the mechanical properties should ideally match the mechanical properties of underlying peripheral nerve, which exhibit Young’s moduli on the order of 1-50kPa. Temporary conformal hydrogel-based electrodes could form high resolution interfaces with peripheral nerves and obviate challenges with chronic inflammatory stimuli. Here, we describe ultracompliant non-biofouling substrate materials for hydrogel-based electronics composed of adhesion-promoting moieties and zwitterionic polymer networks. This class of polymers exhibits anti-biofouling ability and compatibility with transfer printing of electronic structures to hydrogel substrates.
Materials and Methods: Catechol-bearing monomer dopamine methacrylate (DMA) was prepared according the previous report, while the zwitterionic 2-Carboxy-N,N’-dimethyl-N-(2’-(methacryloyloxy) ethanaminium inner salt (carboxybetaine methacrylate, CBMA) was synthesized by the reaction of 2-(N,N’-dimethylamino)ethyl methacrylate (DMAEMA) and t-butyl bromoacetate. Then, the catecol-bearing Hydrogels P(CBMA-co-DMA) were prepared by though UV photocrosslinking with DMA and CBMA (Figure 1a). Fourier transform infrared (FTIR) spectrum of dehydrated gels were recorded with 50 scans at 4 cm-1 resolution from 4000-400 cm-1. The mechanical properties of hydrogels were measured using a rheometer. Adhesion measurements were measured by the hydrogel-coated identer companying with the custom-made software to obtain the force-distance curves. For microstructural electrode fabrication, transfer printing was used through laminating target hydrogels onto the donor substrate, followed by dissolving the donor substrates for delamination (Figure 1b).
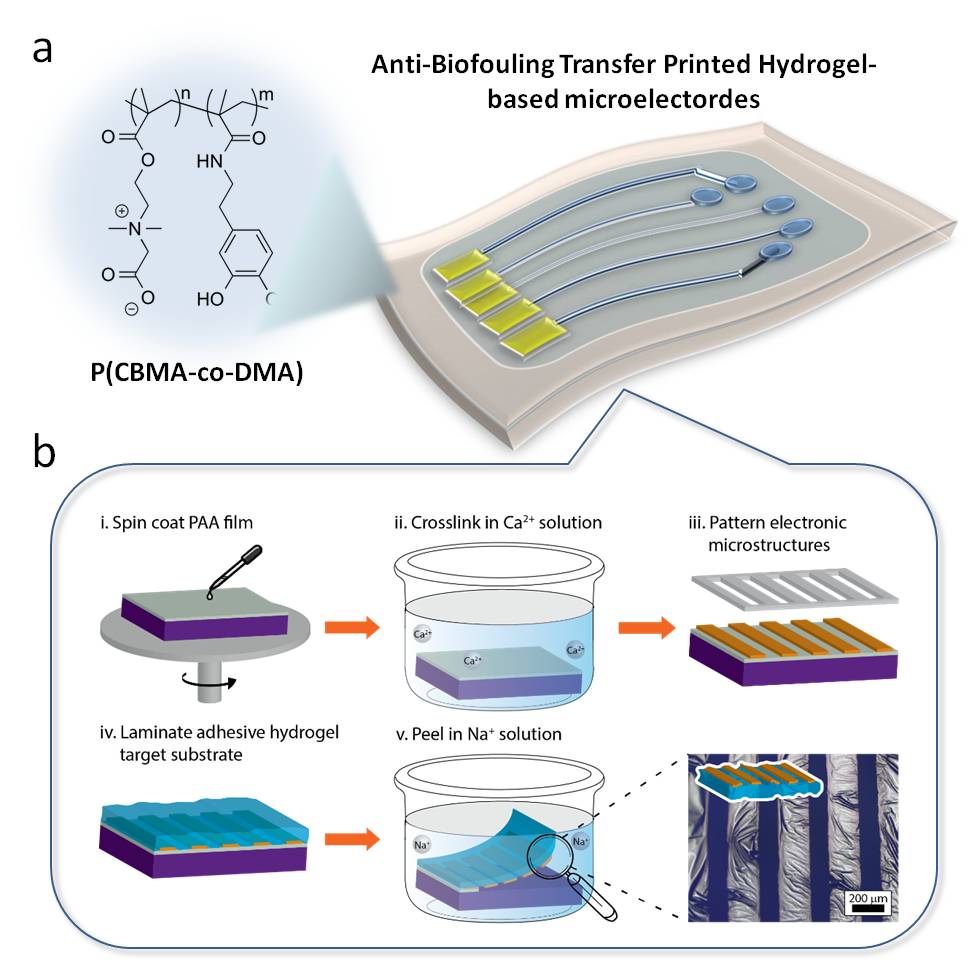
Results and Discussion: By controlling the incorporated ratio of DMA/CBMA, the ability to resist nonspecific protein absorption can be controlled, making the copolymer exhibit gel structure with tunable adhesion intensity without any cell adhesion investigated by in-vitro test (Figure 2a). On the other hand, based on the adhesion contributed by DMA, we directly manufactured the hydrogel into a device by using transfer printing which is a facile way to facilitate patterning electronic microstructures on the swollen hydrogels (Figure 2b and c). Through thin film patterning, deposition techniques, and transfer printing, a hydrogel-based multi-channel microelectrode array integrated with the Pt electrodes in the size of 100μm and the insulation layer of Al2O3/parylene C has been successful developed.
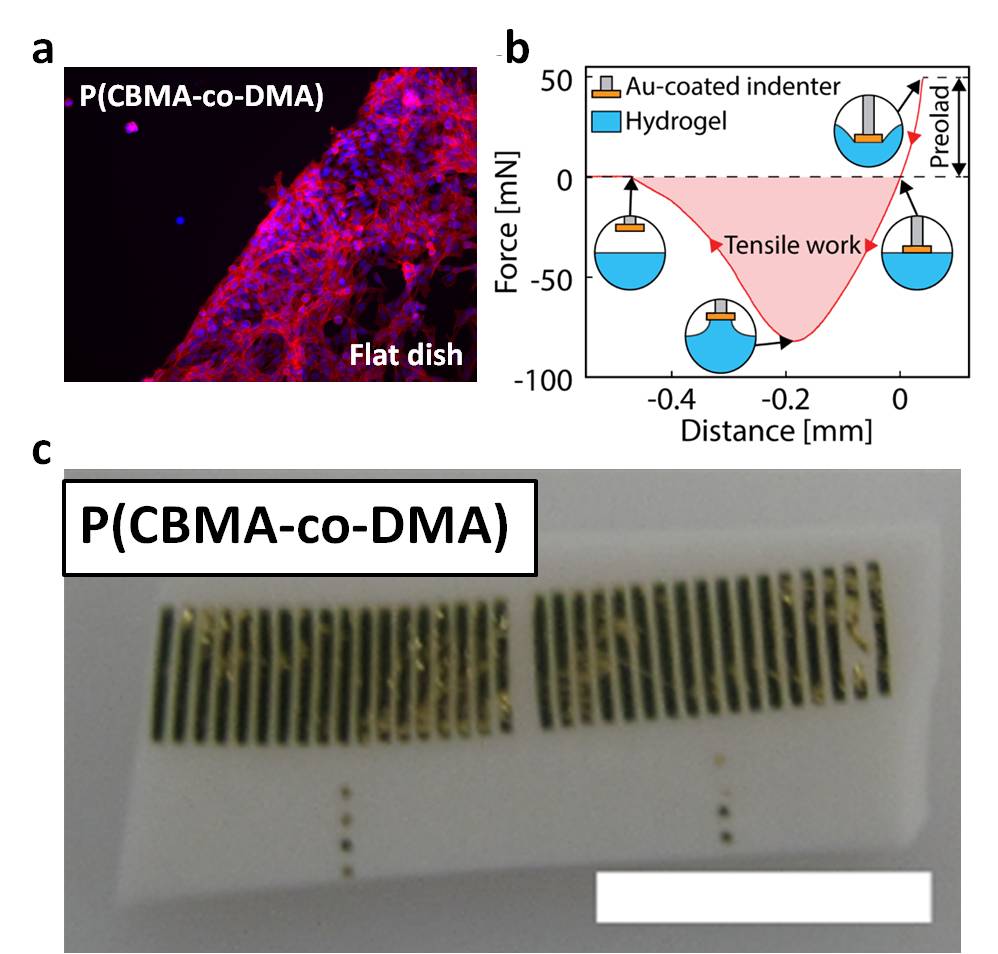
Conclusion: We demonstrate that P(CBMA-co-DMA) catecol-bearing zwitterionic hydrogels exhibits anti-biofouling ability to nonspecific proteins which is recognized to be anti-inflammatory. Also, the hydrogels have robust adhesion to allow the integration of electronic structures by transfer printing. In the mean time, we demonstrate the fabrication strategy composed of zwitterionic swollen hydrogel substrates and microfabrication techniques are highly desired for the in-vivo application to improve the performance of implanted bioelectrodes.