Introduction: Upon biomaterial implantation, macrophages (MΦ) adhere to the material surface and can fuse to form large, multinucleated Foreign Body Giant Cells (FBGCs) [1]. MΦ adherence and FBGC formation on implanted biomaterial surfaces are pivotal events in the progression of a Foreign Body Response (FBR). The host response is further compounded by protease release from FBGCs, during a process described as ‘frustrated phagocytosis’, causing both biomaterial and tissue component degradation in the peri-implant region [2]. Previous studies highlight roles for matrix metalloproteases (MMP)-9 in MΦ fusion [3]. However, little is known regarding the role of serine proteases in FBGC biology. Work presented details the proteolytic activity of FBGCs and effect of serine protease inhibition on the fusogenic capabilities of primary murine MΦs.
Materials and Methods: Fusion assays were performed using primary murine bone marrow derived macrophages (BMM) isolated from WT C57BL/6 mice, and treated with IL-4 (10 ng/ml), GM-CSF (10 ng/ml), and 100 µM affinity-based biotinylated inhibitor probes (or vehicle control) targeting three classes of serine proteases; trypsin-like, chymotrypsin-like and elastase-like proteases [4]. Each inhibitor probe was tested alone, and in combination to highlight possible synergistic activity. Protease temporal expression and activity during FBGC formation was analysed by SDS-PAGE, Zymography and Western Blotting from cell-conditioned media and whole cell lysates.
Results: Increased serine protease secretion in FBGCs at Day 2, 5 and 7 is observed, when compared with non-fused mononuclear controls. These proteases range from 38-49 kDa in size. FBGC formation is reduced by almost 50% when subject to protease inhibitor treatment. Statistical analyses show BMM treatment with inhibitors directed towards trypsin-like (P < 0.01), chymotrypsin-like (P < 0.001) and elastase-like (P < 0.05) activity results in a significant decrease in percentage fusion when compared with controls. Combined treatment with all three protease inhibitors dramatically reduces FBGC formation in vitro.
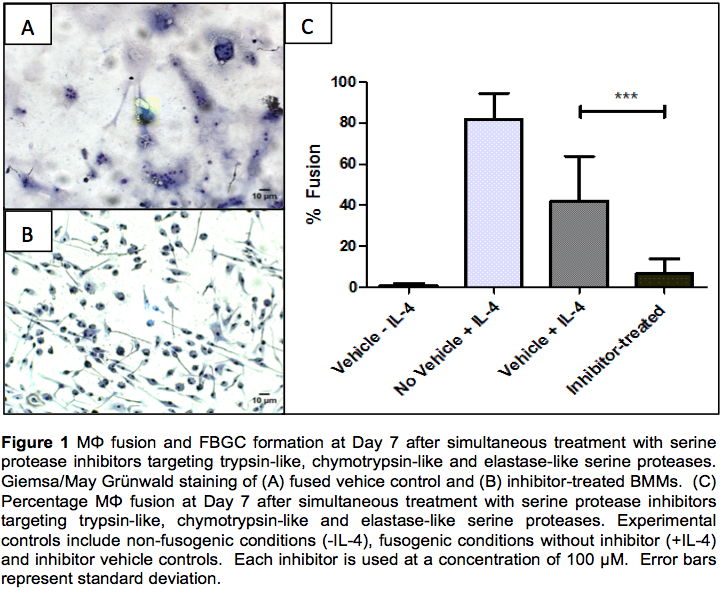
Discussion: Western blotting, ultilizing the biotinylated inhibitor probes, suggests an increase in protease activity in fused BMMs overtime when compared with unfused controls, in particular an increase in trypsin-like and chymotrypsin-like actiivity at Day 5 and 7. Simultaneous treatment with all three serine inhibitors targeting chymotrypsin-like, trypsin-like and elastase-like serine proteases results in a significant reduction in FBGC formation, over and above each inhibitor alone, and is indicative of a synergistic effect. Such treatment represents a viable novel strategy to abrogate macrophage fusion.
Conclusion: Together, these results denote a function for serine proteases in MΦ fusion and FBGC biology. Serine proteases may represent potential novel therapeutic targets in efforts to prevent the FBR – be it due to their influential role initiating intracellular signalling or indeed orchestrating cytoskeletal reorganisation during the fusion process. Future work will centre on the identification and functional characterisation of these serine proteases, unmasking their mechanistic role in FBGC formation.
References:
[1] J.M. Anderson et al, "Foreign body reaction to biomaterials", Seminars in Immunology, Vol. 20, Jan. 2008.
[2] S. Franz et al, "Immune responses to implants - A review of the implications for the design of immunomodulatory biomaterials", Biomaterials. Vol. 32, June 2011.
[3] S. MacLauchlan et al, "Macrophage fusion, giant cell formation, and the foreign body response require matrix metalloproteinase 9", Journal of Leukocyte Biology, Vol. 85, April 2009.
[4] B. F. Gilmore et al, "Expedited solid-phase synthesis of fluorescently labeled and biotinylated aminoalkane diphenyl phosphonate affinity probes for chymotrypsin- and elastase-like serine proteases", Bioconjugate Chemistry. Vol. 20, August 2009